First Cancer Patient Treated with "Base-Edited" T Cells
Earlier this week, Great Ormond Street Hospital for Children (GOSH) in London released an inspiring statement regarding a young patient named Alyssa, who received a revolutionary cancer treatment consisting of “base-edited” T cells.
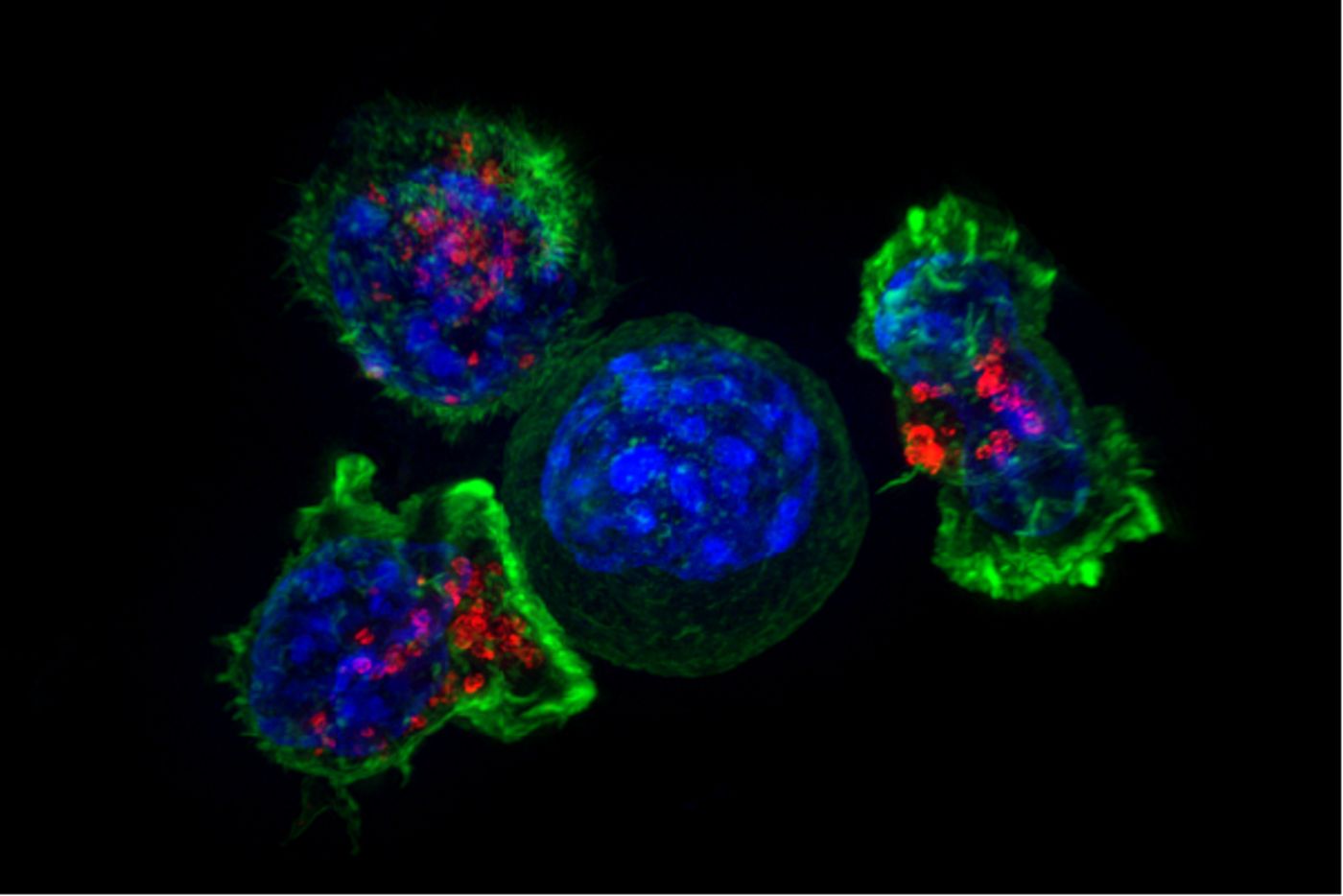
T cells, a subset of immune cells integral in killing cancer cells, have gained significant attention in the past decade due to the success of immunotherapies. One of the more recent immunotherapeutic advances involves CAR T cell therapy which can eradicate advanced cases of blood cancers like leukemia. CAR T cell therapies remain a unique treatment option as oncologists and scientists specially engineer personalized cells for each patient.
Doctors first collect T cells from the patient to generate CAR T cells. Then, in a laboratory setting, the doctors engineer the cells to display proteins called chimeric antigen receptors (or CARs) on their surface. These specially engineered CARs recognize proteins found on the patient’s cancer cells.
According to the press release, Alyssa became the first patient in the world to receive a special form of CAR T cell therapy generated from the T cells of a healthy donor. Alyssa, now 13, was first diagnosed with T-cell acute lymphoblastic leukemia (T-ALL) in 2021. After standard therapies, including chemotherapy and a bone marrow transplant , failed to eradicate her cancer, Alyssa entered a clinical trial in May 2022 at GOSH. Just one month after receiving base-edited CAR T cells, Alyssa entered remission, and she has remained cancer-free throughout subsequent monitoring.
So, how does the revolutionary treatment Alyssa received differ from traditional CAR T cell therapy? In short, it protects the specially designed CAR T cells from attacking themselves, a process known as fratricide. Because T-ALL is characterized by abnormal T cells, developing a CAR to recognize the cancerous T cells and spare the re-engineered T cells poses a significant challenge. This principle also explains why most successful CAR T cell therapies treat B cell malignancies but not T-ALL like Alyssa’s cancer.
To circumvent the challenges facing CAR T cells in T-ALL, the researchers at GOSH and their collaborators at UCL Great Ormond Street Institute of Child Health (UCL GOS ICH) utilized a new gene editing strategy called base-editing. This technique chemically alters the genome sequence of the T cells. By altering a single base in the genetic code, the instructions the genome provides the body change.
GOSH summarized the gene edits undertaken to generate the CAR T cells used for Alyssa’s treatment. First, the researchers re-engineered the cells obtained from the healthy donor so that the patient’s immune system would not recognize them as foreign and eliminate them. Next, the procedure removed two “flags” from the T cells to ensure that the CAR T cells, or other cancer therapies, would not attack them. The final genetic modification guaranteed that the modified T cells recognized cancerous malignant T cells.
The researchers also used their gene-editing strategy on T cells collected from healthy donors, sparing Alyssa, or other patients like her, from undergoing the T cell collection procedure.
While Alyssa is just the first pediatric T-ALL patient treated with this gene editing strategy, it can revolutionize cancer treatment, providing patients with limited therapeutic options with a new therapy.
Sources: GOSH News, Nature (Melenhorst), Blood, J Hematol Oncol, Nature (Komor), BBC News, NCI Visuals Online