Fighting Melanoma without T Cell Activation
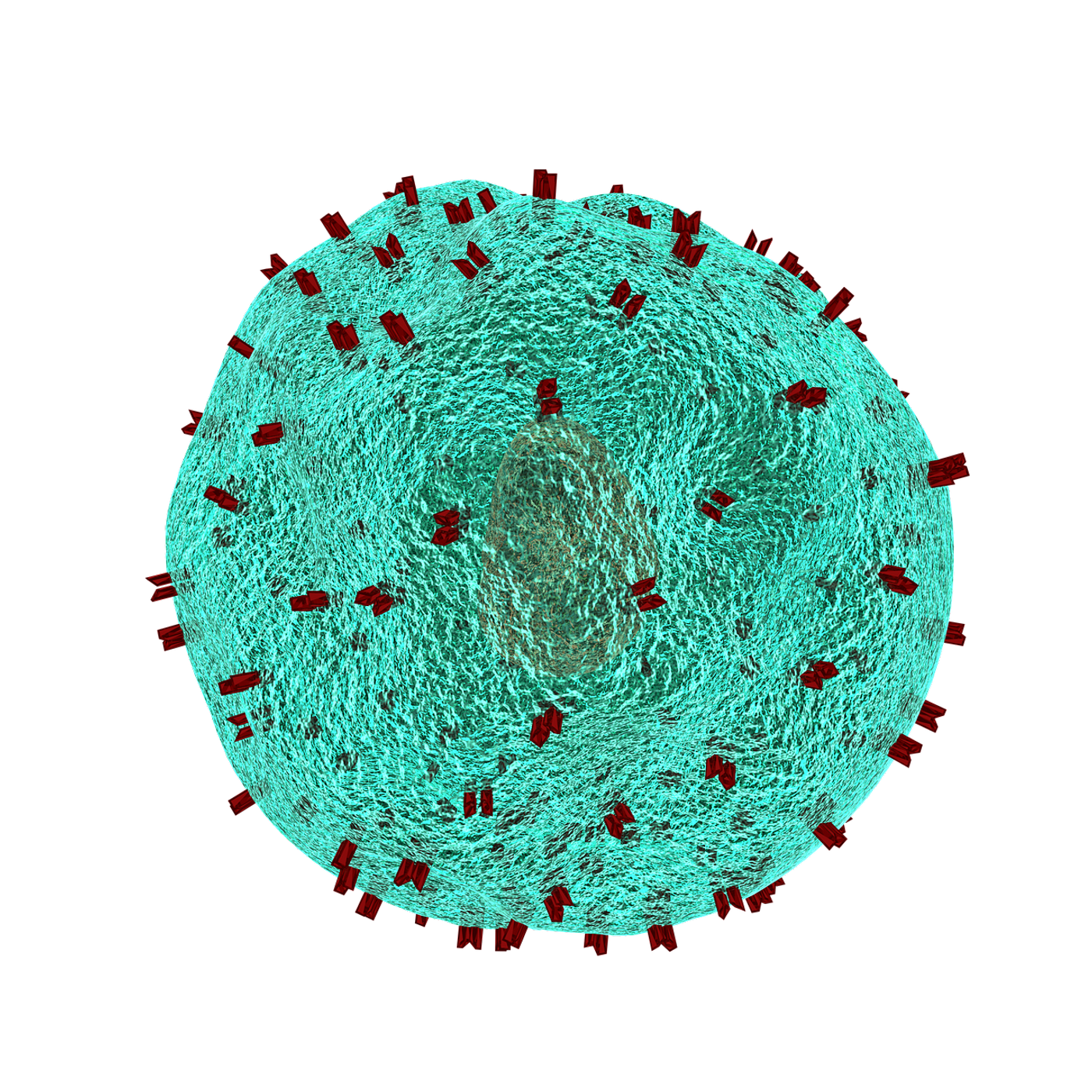
Cytotoxic T cells (CTLs), a type of immune cells integral to fighting cancer, rely heavily on interaction with a molecule called major histocompatibility complex (MHC) class I. Found on the surface of cells, MHC class I molecules display pieces of protein that have been degraded inside the cell. When a MHC class I molecule binds to a T cell receptor (TCR) on the surface of a CTL, the immune system activates. Once activated, CTLs attack cells displaying an abnormal protein, such as cancer cells. Because of this important function, CTLs are also called “killer” T cells.
MHC class I molecules contain a protein called beta-2-microglobulin (β2M), which facilitates the presentation of tumor peptides to CTLs. β2M deficiencies, which can occur due to genetic mutations, prevent CTLs from recognizing cancer cells. Loss of β2M also hinders susceptibility to immunotherapies, including immune checkpoint inhibitors (ICIs). However, in some cases, β2M-inactivated tumors do respond to ICI. This presents a question regarding how the immune response activates without sufficient β2M and MHC class I. A team of researchers sought to interrogate this question and recently published their findings in Cancer Immunology Research.
The researchers first knocked out β2M in different kinds of mouse cancer cells. Each cancer cell expressed different levels of MHC class I and thus varying susceptibility to the ICI anti-programmed death-1 (αPD-1). Two cell lines, MC28 colorectal cancer cells and YUMMER2.1 melanoma cells, still responded to αPD-1 despite lacking β2M. The study demonstrated a dependency of αPD-1 response on two types of immune cells, CD4+ T cells and natural killer (NK) cells.
Next, the researchers looked at biopsies from melanoma patients before they received αPD-1 treatment. While they rarely detected samples with a complete loss of β2M, the analysis revealed that many biopsies lacked one of the two copies of the gene for β2M, a condition called loss of heterozygosity (LOH). Notably, the data showed an enrichment of β2M LOH in the biopsies of patients who failed to respond to αPD-1 therapy. Further, the same samples with high levels of β2M LOH were highly infiltrated with NK cells.
The authors conclude that, in mouse cancer models, activation of CD4+ T cells and NK cells facilitates responsiveness to PD-1 blockade in the absence of β2M. Further, NK cells likely play a similar role in retaining αPD-1 sensitivity in human melanoma with partial β2M loss.
Sources: Front Immunol, NEJM, JCO Precision Oncol, Cancer Immunol Res