Researchers Successfully 'Transplant' Mitochondria From One Cell to Another
Our cells are full of little machines that perform certain critical functions, like ribosomes, which help generate proteins, or mitochondria, the powerhouses of the cell. Now scientists have developed a method that uses a nanosyringe to transplant mitochondria from one cell to another. This technique may be one way to rejuvenate damaged cells, like when organs are replaced. The findings have been reported in PLOS Biology.
It's thought that mitochondria were once tiny microbes that infected another cell long ago, and took up residence there. Those organisms developed an endosymbiotic relationship, and the mitochondrial organelle was born, which helped give rise to the complex organisms, including plants, fungi, and animals, which are made of eukaryotic cells. Since mitochondria still carry their own genomes and even produce their own proteins, there is real evidence that this occurred during evolution.
In human cells, there isn't only one mitochondria; there are many, which create a network of threads. Those threads can respond to pressure, "and transform into something like a string of pearls, from which individual mitochondria subsequently pinch off," explained lead study author Christoph Gäbelein.
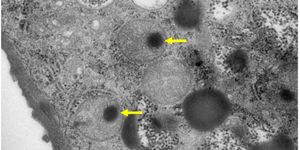
Using a nanosyringe they created for this method, the researchers were able to pierce through the membrane surrounding a cell, and pick up individual mitochondria. Then, they punctured the membrane of a different cell, and placed those mitochondria inside.
Gäbelein noted that the procedure is minimally invasive, and both cells survive the process. A laser generated by a converted atomic force microscope controls the position of the nanosyringe. The pressure of the syringe is regulated and adjustable, and during the transplants it can transfer femtoliters, an extremely tiny amount of fluid as small as a millionth of a millionth of a milliliter.
Over 80 percent of the mitochondria withstood the process as well. Although some mitochondria were targeted by the recipient cell's quality control machinery, and destroyed, the researchers observed most mitochondria fusing with the existing network in the recipient cell within twenty minutes of transplantation.
"The host cell accepts them," said study leader Julia Vorholt of the Institute of Microbiology at ETH Zurich.
The study authors suggested that this technology could be applied in various ways. It may be possible to boost the health and function of stem cells that are showing signs of metabolic decline, for example.
The researchers are also now interested in investigating "the processes that control how different cell compartments cooperate, and we hope to unravel how endosymbioses develop over evolutionary time," Vorholt said.
Sources: ETH Zurich, PLOS Biology