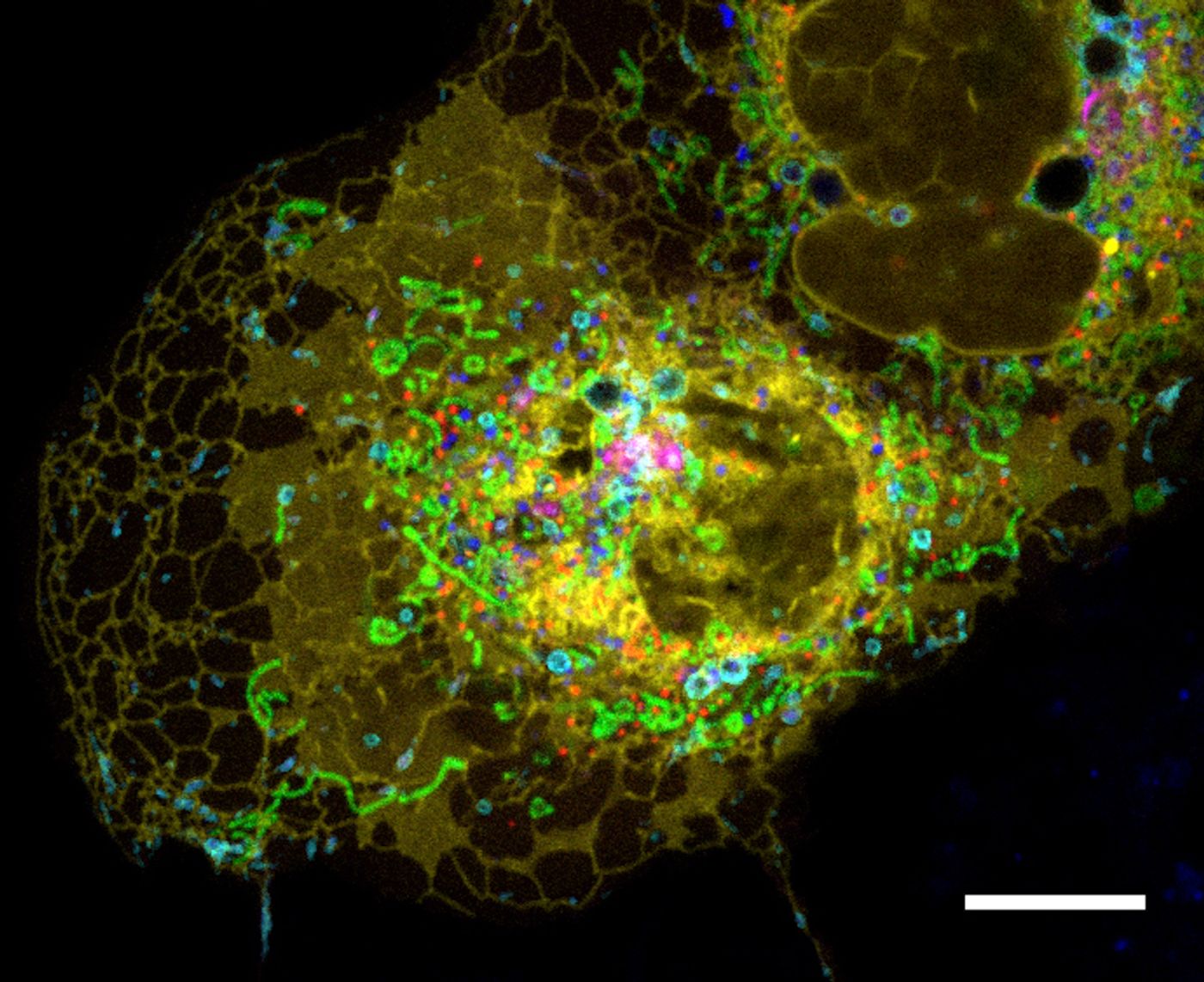
The lysosome is a small membrane bound organelle that can be found in most animal cells. It was once thought that these little sacs were like recycling centers that could take up cellular waste products, and digest them with the enzymes inside, which freed up stud like lipids, sugars, or amino acids to be reused by the cell. Many other crucial functions have recently been found for lysosomes, including roles in immunity signaling and nutrient sensing, for example. Dysfunction in lysosomes has also been linked to a variety of human diseases.
Now, researchers have developed a technique that can identify all of the molecules in the lysosomes of a mouse model. This work will hopefully help provide insights into how lysosomal disorders are caused, and how they might be treated.The work, which has been reported in Nature, has also shed light on a neurodegenerative disorder called Batten disease.
“Lysosomes are fascinating both fundamentally and clinically: they supply the rest of the cell with nutrients, but we don’t always know how and when they supply them, and they are the places where many diseases, especially those that affect the brain, start,” noted study leader Monther Abu-Remaileh, an assistant professor at Stanford University.
Lysosomal storage disorders are a group of diseases that occur when there are genetic mutations in molecules that are typically found in lysosomes. Studying these disorders has proven challenging, however. Researchers often disrupt a protein they are interested in to see what happens to the cell after the protein is disrupted. But huge increases in the levels of molecules that sit inside of lysosomes are difficult to detect, because they get "buried in the noise" created by the rest of the cell, noted first study author Nouf Laqtom. “If something happens and a molecule grows in abundance 200-fold in the lysosome, you would see only a two-fold increase if you look at the whole cell.”
By isolating lysosomes from the rest of the cell, researchers could quiet that noise. To do so, they genetically engineered mice that would carry molecular tags on their lysosomes. Any tissue in the mice could be collected, then the lysosomes could be isolated from that tissue by using the tag. The molecular tags on the lysosomes could be bound by matching them to molecular clamps that the researchers attached to little magnetic beads. A second magnet could be applied to a tissue slurry containing those linked lysosomes, and all of the lysosomes were extracted with the magnet.
Once the lysosomes were isolated, a tool called mass spectrometry was applied. This identified the molecular weights of the molecules that had been isolated from the lysosomes, which gives clues to their identities and their functions.
These mice can now be used by the research community at large. Some groups have already started to work with this model, called the LysoTag mice.
“We hope that this will become the gold standard,” said Abu-Remaileh.
Once the method was developed, the researchers and colleagues investigated the impact of a mutation in the gene CLN3, which encodes for a protein on the surface of lysosomes. This mutation has been linked to juvenile Batten disease, a fatal disorder that causes seizures, and neurodegeneration in children and young adults.
The researchers found that there was a significant increase in molecules called glycerophosphodiesters (GPDs) in the mouse model. Normally, GPDs would not build up in lysosomes; they would move to another part of the cell and get degraded. But the scientists hypothesize that CLN3 is involved in helping GPDs get to where they are going. When CLN3 doesn't do that work, the GPDs accumulate. GPDs were found in the cerebrospinal fluid of patients. Now, the scientists want to know if GPDs may be toxic and how drugs may help shuttle them.
Sources: Stanford University, Nature