Changes in Protein Folding Can Drive Evolution
In cells, protein-coding genes are transcribed into messenger RNA (mRNA) molecules when they are active, and mRNA can be translated into proteins. While protein start out as strings of amino acids, they are folded into three-dimensional structures to carry out their function, and when they are misfolded, serious problems can arise. However, new research has suggested that changes in protein folding also seem to drive evolution. The findings have been reported in Science Advances.
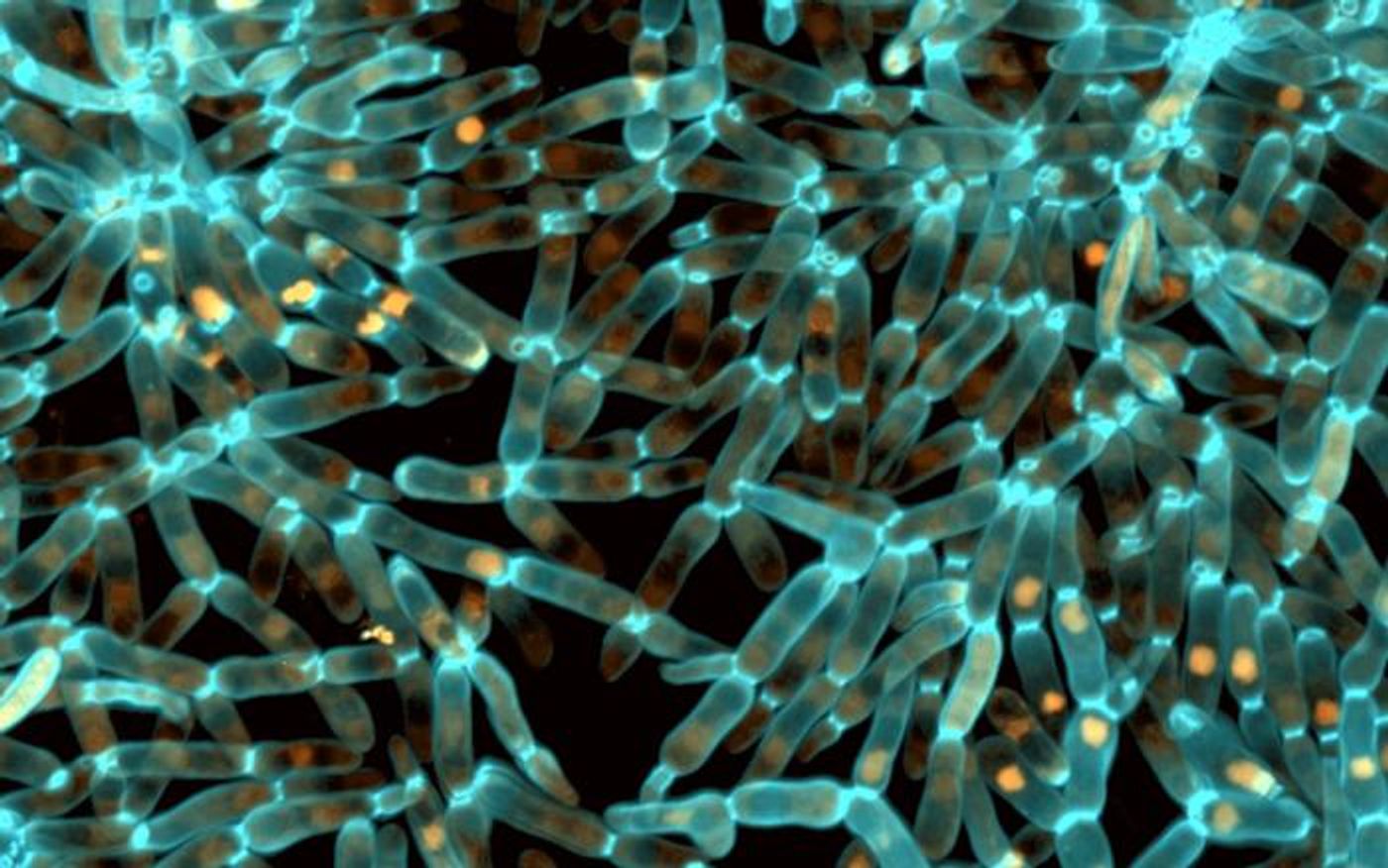
In this work, the investigators turned to a model of evolution, an ongoing project called the Multicellularity Long Term Evolution Experiment (MuLTEE). Snowflake yeast are being used in MuLTEE to research the emergence of novel features and functions in multicellular organisms.
This research has highlighted the ability of protein changes to facilitate evolution, and why the genetic code cannot fully explain how adaptive behaviors arise in organisms, explained co-corresponding study author Associate Professor Juha Saarikangas of the University of Helsinki. "Achieving such understanding requires mapping the entire flow of genetic information, extending all the way to the actionable states of proteins that ultimately control the behavior of cells."
Scientists have been able to observe snowflake yeast evolving over 3,000 generations in MuLTEE. These organisms started out with cells that were weaker than gelatin, and they eventually became as tough as wood.
The researchers wanted to know how this phenotype evolved, and found that it involved protein folding. A chaperone protein called Hsp90 aids in the formation of other proteins, and it was found to be expressed at lower levels in tougher, larger snowflake yeast cells compared to weak cells. Hsp90 was acting like a dial, and disrupting a crucial molecule that helps control cell division. This led to the development of lengthier yeast cells that began to wrap around each other and form bigger, tougher, multicellular groups.
Scientists have known that Hsp90 aids in protein folding and stabilization. "What we've found is that slight alterations in how Hsp90 operates can have profound effects not just on single cells, but on the very nature of multicellular organisms," noted first study author Kristopher Montrose of the Helsinki Institute of Life Science.
"We tend to focus on genetic change and were quite surprised to find such large changes in the behavior of chaperone proteins. This underscores how creative and unpredictable evolution can be when finding solutions to new problems, like building a tough body," added co-corresponding study author Professor Will Ratcliff of the Georgia Institute of Technology.
Sources: University of Helsinki, Science Advances