A magnetic bead for every need: Expanding applications with conjugated magnetic beads
Magnetic beads (or superparamagnetic particles) can be used for the isolation and purification of cells, nucleic acids, proteins or any other biomolecules. The surface of magnetic beads can be coated in a variety of ligands to bind a specific target molecule. By exploiting this type surface modification, magnetic beads offer an exceptionally versatile tool for biomolecule isolation.
In this article, we discuss how the functionalization of the surfaces of magnetic beads through ligand conjugation massively extends their potential in life sciences, simplifying and improving consistency in a range of applications.
Discover our broad portfolio of magnetic beads
A typical magnetic bead workflow
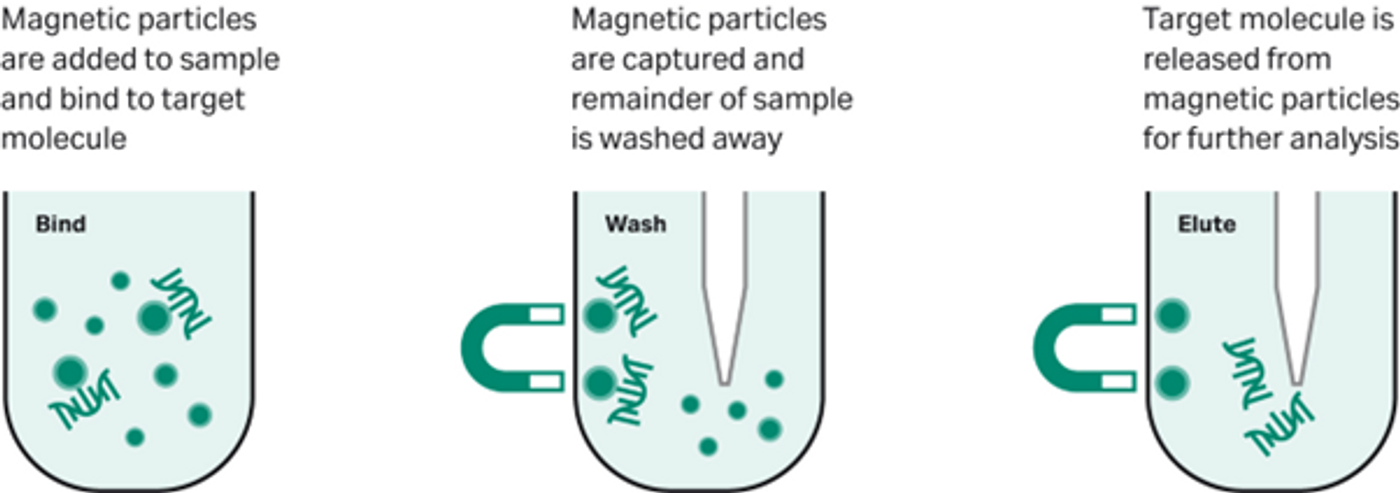
Magnetic separation of biomolecules is a well-established alternative to centrifugal and column-based approaches. Briefly, these beads are iron-oxide particles encased in a coating with a biological or chemical agent known to bind a specific target.
When these beads are placed in solution, they capture the target biomolecules on their surface. Upon applying an external magnetic field (usually by placing in a magnetic rack), the beads are attracted to the magnetic source on the outer edge of the sample tube and aggregate together, separating the bound targets from the bulk solution (Fig 1).
Fig 1. A typical magnetic bead-based workflow.
Magnetic bead conjugation: How does it work?
By altering the surface properties of magnetic beads – commonly known as surface functionalization – it is possible to be highly specific about the types of biomolecules the beads will bind. Achieved by conjugating a specific ligand suited for the binding or functionality needs, magnetic beads offer an extremely flexible platform that can be tailored for specific applications.
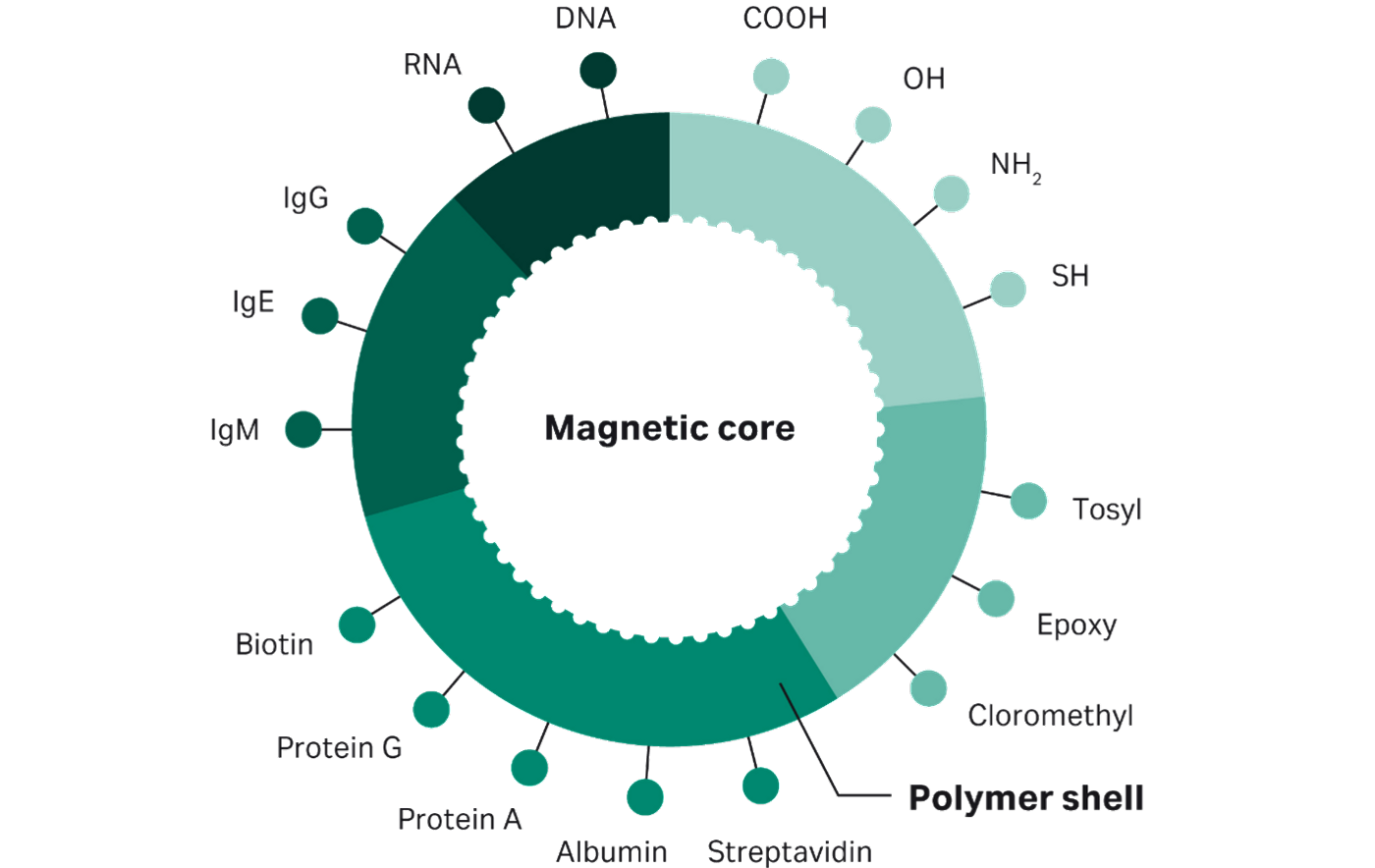
Magnetic beads can be conjugated with a wide variety of ligands due to the variety of different composition, surface chemistry, and size possibilities of beads. Ligands can be bound covalently and non-covalently and link either directly to the bead surface or indirectly through a reactive intermediate. Figure 2 shows a few possible functionalization examples including common functional groups, proteins, antibodies and nucleic acids.
There are several possible approaches to conjugating a ligand to a magnetic bead. Typically, bead surfaces are first functionalized, which involves coating the beads to present a functional group, enabling the attachment of a specific ligand. Then, the ligand which will bind specifically to the molecule of interest can be conjugated to the functional coating.
Two common functional surface chemistries used in conjugation applications include: carboxylate-modified and amine-blocked magnetic beads.
Modified surfaces
Magnetic bead surfaces modified with functional groups can facilitate the attachment of ligands by both adsorption and covalent bonds. For example, proteins are rapidly adsorbed onto the surface of carboxylate-modified particles, mediated by hydrophobic and ionic interactions.
While this method is simple, the relatively weak interactions can result in challenges of instability. Additionally, the final orientation of the ligand when adsorbed onto the surface cannot be specified, potentially leading to an increase in non-specific binding.
To overcome stability and specificity problems, proteins can also be covalently attached to the surface of, for example, carboxylate-modified magnetic beads. Taking this approach requires first activating the carboxyl groups with water soluble carbodiimide 1-ethyl-3-(3-dimethylaminopropyl) carbodiimide (EDAC); which reacts with free amino groups of the adsorbed protein to form amide bonds. This chemistry can be utilized in either a quick one-step protocol or as a two-step process for better control.
Pre-activated surfaces
Pre-activated surfaces – such as tosyl, epoxy, and chloromethyl groups – simplify things further by allowing for very stable covalent coupling without the need for an activation step. In turn, attachment protocols are typically straightforward, requiring a single incubation step in the correct buffer at the appropriate temperature and pH.
Both the chemical group and the incubation conditions will dictate what can be bound. For example, at neutral pH, tosyl groups will bind to sulfhydryl groups whereas a more basic pH is needed for binding amino groups. Similarly, epoxy groups will bind thiol groups at a slightly basic pH and amino groups under higher pH conditions.
This pH-dependent binding provides a useful level of flexibility binding ligands to pre-activated surfaces with the caveat that binding might have less specificity compared to the modified surfaces.
Expanding applications with conjugated magnetic beads
Magnetic beads can now be conjugated with an exceptional variety of ligands ranging from antibodies, antigens, peptides, and proteins to enzymes, nucleic acids, and carbohydrates. Consequently, it is possible to target an extraordinary diversity of biomolecules and, therefore, applications.
Indeed, conjugated magnetic bead systems have been utilized for everything from benchtop research, drug screening, and diagnostics to food production and wastewater treatment. Additionally, the ability to couple magnetic beads with various biosensing devices and microfluidic systems adds further possibilities to the miniaturization of detection and separation technology.
Diagnostics: protein- and antibody-conjugated magnetic beads
Potentially one of the most exciting applications for conjugated magnetic beads is diagnostics. Broadly, by concentrating, isolating, and/or purifying various target analytes, high quality samples containing concentrated target molecules can be made readily available for diagnostic analysis.
For example, the concentration and purification of pathogenic microorganisms by antibody-conjugated magnetic bead separation circumvents the need for laborious and time-consuming cell enrichment processes like bacterial cell culture.
Magnetic beads can also be used more directly in diagnostics. For instance, magnetic immunoassay (MIA) is a type of diagnostic immunoassay that exploits magnetic beads for the detection of a specific analyte.
In MIA, either an antigen or antibody is conjugated to a magnetic bead. If an antibody is paired with the magnetic bead, its corresponding antigen (e.g., virus, toxin, bacteria) will bind when the magnetic bead is placed in the sample. A magnetic reader can then measure the magnetic field change induced by the beads and, since the signal is proportional to the analyte concentration in the sample, be used to give a quantitative diagnosis.(1)
This system offers several advantages over traditional methodologies such as ELISA or Western blot. Primarily, MIA is conducted in a liquid medium whereas ELISA and Western blots require a stationary medium to bind the desired target. A liquid medium-based detection system allows measurement within the model system and, in turn, can provide a more accurate result.
As a final example, immunoprecipitation (IP) of proteins from cell lysates is a common method used to enrich and purify proteins for research and diagnostics. Protein A, Protein G, and Protein A/G are commonly used in IP experiments as they specifically recognize IgG antibodies, an important and common serum antibody in animals.
Traditional IP protocols targeting IgG employ a combination of agarose beads conjugated to either Protein A, G, or A/G and centrifugation. However, agarose and sepharose beads with a magnetic core — such as our Protein A Mag Sepharose™ Xtra beads — offer a simple and efficient alternative that generally gives lower background binding than agarose beads.
As an example of this, Burbelo et al. (2017) demonstrated how magnetic beads modified with protein A/G could bind antibody-luciferase-labeled antigen complexes to simply and rapidly measure specific antibody levels in a variety of infectious and autoimmune disorders.(2) In the case of Sjögren’s syndrome, saliva samples were taken and only required approximately one minute per assay, showing the potential of this type of test at point-of-care.
Drug discovery and screening: Enzyme-conjugated magnetic beads
Enzymes are a common drug target since they are highly susceptible to inhibition by low molecular weight, small, drug-like molecules. However, identifying potential drug targets for a given enzyme requires lengthy, complex, and expensive pharmacological assays.
Additionally, while the majority of approved drugs are derived from natural origins, therapeutically active components in natural products are often found at extremely low concentrations. Attempting to isolate or even just detect these potentially useful compounds is difficult, if not impossible, with traditional separation and analytical methods.
Affinity-based screening approaches circumvent the need to separate potentially important compounds from a complex mixture and can be instead analyzed as a whole. Enzyme-conjugated magnetic beads are beginning to show great promise in affinity-based screening, allowing quick separation and identification of active components from complex mixtures. Additionally, they can be integrated with mass spectrometry and liquid chromatography to help clarify the structures of these active components.
For example, neuraminidase — an enzyme proposed as a potential therapeutic target for influenza — was immobilized onto the surface of magnetic beads to screen for potential inhibitors.(3) Based on enzymatic activity, the researchers were able to identify four compounds as inhibitory ligands of neuraminidase from a botanical matrix.
In other applications, enzyme immobilization onto magnetic beads offers a convenient way to control and reuse enzymes. In addition, magnetic beads can improve the performance of enzyme analytical assays as they provide a relatively large number of binding sites, resulting in improved enzyme kinetics.(4)
Detection of specific sequences: Oligonucleotide conjugated magnetic beads
Quick, simple, and efficient methods for the detection of DNA and RNA in clinical, environmental, and research settings are extremely desirable. Not only can detection of specific fragments of DNA or RNA be used to diagnose disease, but it can be employed to identify contamination of food and water or help answer research questions.
Magnetic beads have become an important part of many next generation sequencing (NGS) workflows, where total DNA and RNA is captured or DNA or RNA is selected based on size. However, it is also possible to conjugate specific oligonucleotide probes to magnetic beads to facilitate the detection of target DNA or RNA.
The sandwich hybridization DNA or RNA capture technique can be coupled with magnetic beads to generate a rapid DNA and RNA detection method. Firstly, oligonucleotide probes that are complementary to the target DNA or RNA are designed and synthesized. One probe is conjugated to a magnetic bead and the other may be conjugated to a signal molecule, such as a fluorescent tag. When the conjugated probes are mixed with a sample, any target DNA or RNA will hybridize. Subsequently bound target DNA/RNA can be isolated using magnetic separation and the fluorescent signal measured to confirm its presence.
Catalog or custom?
We offer many pre-conjugated beads, from Oligo(dT)-coated magnetic particles for mRNA isolation and extraction to streptavidin-coated magnetic beads for biotinylated target molecules. However, for specific applications and/or unique ligands, we provide comprehensive custom conjugation services.
Guided by customer requirements, our custom services team develops and manufactures magnetic beads conjugated to meet customer specifications, coupling a specific peptide protein, antibody, or oligonucleotide as well as a fluorescent dye to the surface of high-quality Sera-Mag™ beads.
Find out more about our custom conjugation services
Related content:
- The scientist’s guide to magnetic beads
- Magbeads 101: A guide to choosing and using magnetic beads
- SeraSil-Mag™ silica particle for optimal binding when DNA sample is scarce
- Test your knowledge of magnetic beads
- Webinar: Broaden your applications with magnetic bead conjugation
References:
- Rettcher, S. et al. Simple and Portable Magnetic Immunoassay for Rapid Detection and Sensitive Quantification of Plant Viruses. Appl. Environ. Microbiol. 81, 3039 (2015).
- Burbelo, P. D. et al. Ultrarapid Measurement of Diagnostic Antibodies by Magnetic Capture of Immune Complexes. Sci. Reports 2017 71 7, 1–11 (2017).
- Zhao, Y. M. et al. Magnetic beads-based neuraminidase enzyme microreactor as a drug discovery tool for screening inhibitors from compound libraries and fishing ligands from natural products. J. Chromatogr. A 1568, 123 (2018).
- Sassolas, A., Hayat, A. & Marty, J. L. Immobilization of enzymes on magnetic beads through affinity interactions. Methods Mol. Biol. 1051, 139–148 (2013).