Are You Using the Right Immunoassay for Your Application?
Immunoassays are methods for detecting and quantifying analytes within a biological sample based on specific antigen-antibody reaction. They have been widely used in scientific research and pharmaceutical drug discovery for decades and they can be used to detect a variety of targets, including proteins, lipids, and nucleic acids, as long as there are known antibodies that bind specifically to that target.
Traditional immunoassays involve labor-intensive, time-consuming protocols that require multiple wash and incubation steps. However, the advancement of new detection technologies has led to the development of faster, no-wash immunoassays. In this article, we explore the limitations of common commercially available immunoassay kits with the benefits of new no-wash alternatives and discuss five key questions to consider when selecting the right immunoassay for your research.
Common Types of Immunoassays
Enzyme-Linked Immunosorbent Assay (ELISA)
ELISAs are assays for detecting specific target antigens by immobilizing them to a solid surface, such as a 96- or 384-well polystyrene plate. There are a variety of ELISA methods, but they all involve the following steps:
- Coating: Attaching a specific antigen/antibody to a solid surface
- Probing: Incubation of antigen with antigen-specific primary antibody
- Washing: Removal of the free unbound antigen and antibody
- Detection: Measuring the signal generated from enzyme-labeled primary or secondary antibody
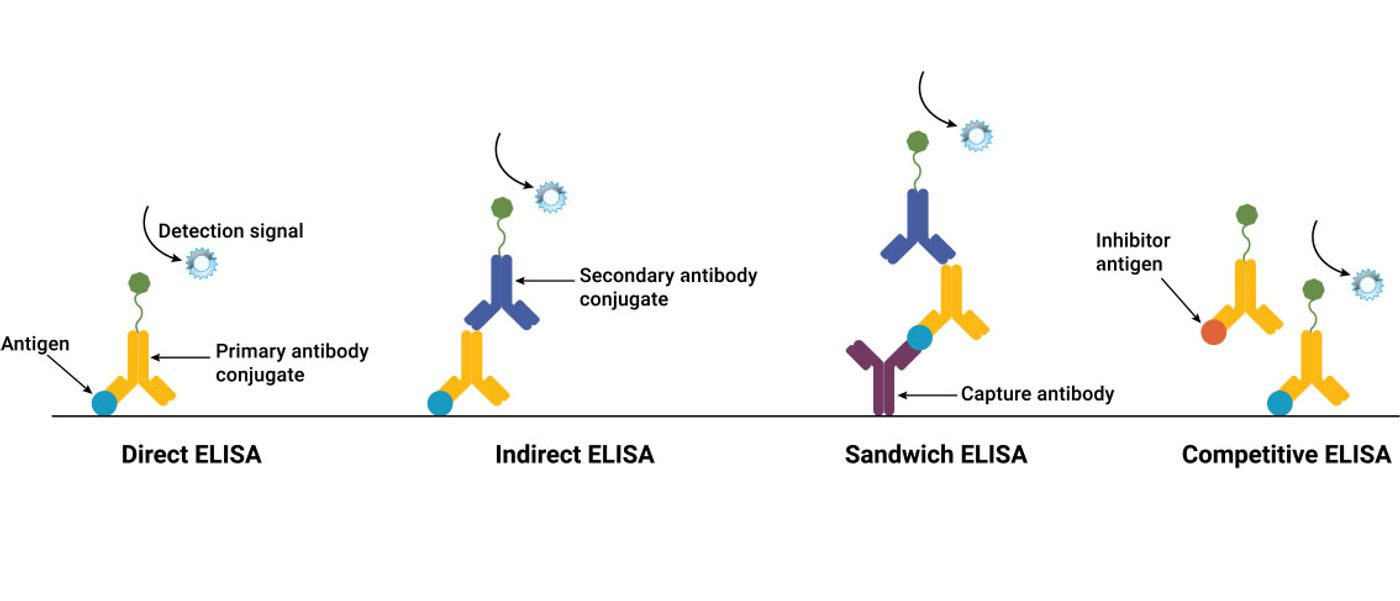
- Direct ELISAs
In direct ELISAs, the antigen is attached directly to the plate surface and incubated with just a primary antibody labeled with a detection label. Signal is generated directly from the primary antibody.
- Benefits: Fewer steps; Faster
- Limitations: Less specific; high background; no signal amplification
- Indirect ELISAs
In indirect ELISAs, the antigen is attached directly to the plate surface. However, unlike direct ELISAs, detection requires incubation with both a primary antibody and secondary antibody. Signal is generated from the secondary antibody conjugated to the detection label.
- Benefits: Allows signal amplification
- Limitations: More steps; potential cross-reactivity from secondary antibody
- Sandwich ELISA
Sandwich ELISAs require two primary antibodies against different epitopes of the target antigen. One of the primary antibodies is immobilized to the plate surface and captures the target antigen. Then, a second primary antibody also binds the target antigen, forming a sandwich complex. Signal is generated from the second primary antibody with the detection label.
Traditional sandwich ELISAs require multiple wash steps. However, some newer commercial “Fast ELISA” kits allow the antibody-antigen sandwich complex to form in one step, therefore requiring only one wash step. The downside to these quick ELISA kits is that they require the purchase of plates precoated with antibodies conjugated to specific affinity tags.
- Benefits: High specificity; high sensitivity
- Limitations: Difficult to find suitable antibody pairs
- Competitive ELISA
Competitive ELISAs are based on competitive binding: a reference antigen or antibody competes with an unlabeled sample antigen or antibody for the same binding site. All of the ELISAs discussed above can be adapted to a competitive format. In one example, a reference antigen is precoated to the plate surface, then sample antigen mixed with labeled antibody is added. During incubation, the sample and reference antigen compete to bind to the labeled antibody. The higher the sample antigen concentration, the fewer labeled antibodies will be bound to the reference antigen, and the lower the detection signal.
- Benefits: Flexibility in format; less sensitive to sample dilution and sample matrix; can use crude sample (no antigen purification required)
- Limitations: Depends on format
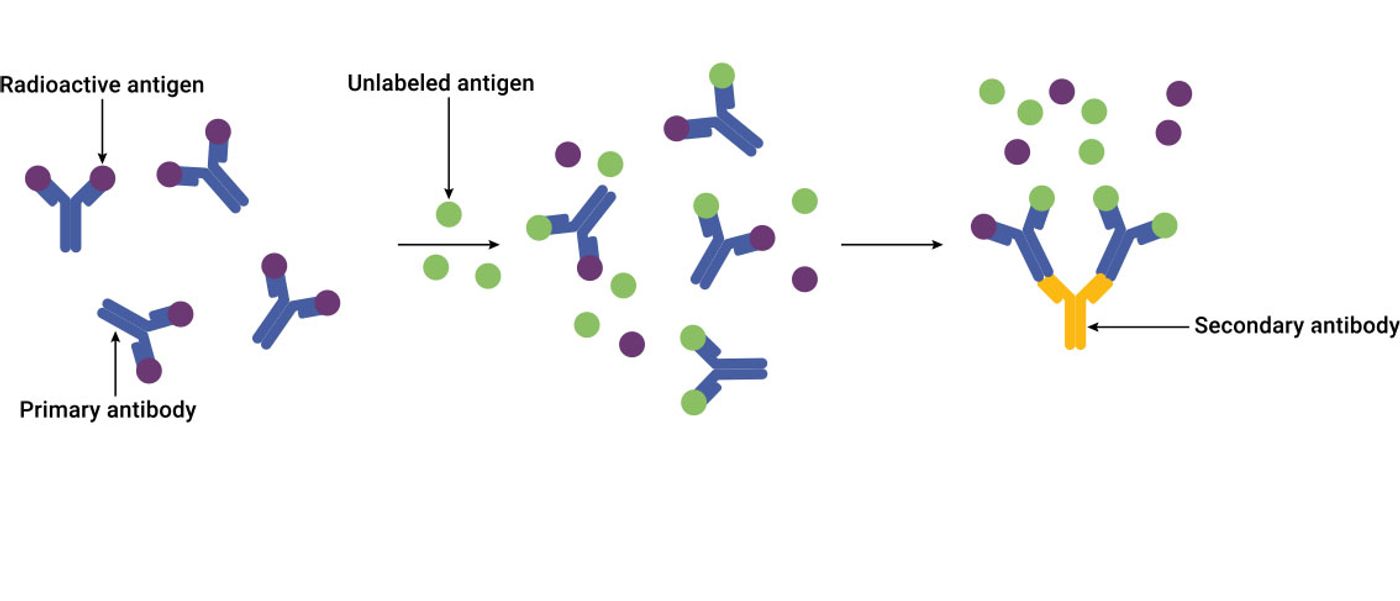
Radioimmunoassay (RIA)
Radioimmunoassays use radioactive isotopes as the detection label. These assays are based on competitive binding. First, a radioactively labeled antigen (called the “tracer”) is mixed with antibody to form antigen-antibody complexes. Then, sample containing unlabeled antigen is added. The unlabeled antigen competes for the same antibody binding sites to displace the tracer (“free tracer”). The radioactive signal of free tracer and antibody-bound tracer is used to measure the concentration of unlabeled antigen within the sample. Less bound tracer and more free tracer means a higher concentration of antigen.
- Benefits: High sensitivity
- Limitations: Safety hazard due to risk of radiation exposure; disposal of radioactive waste
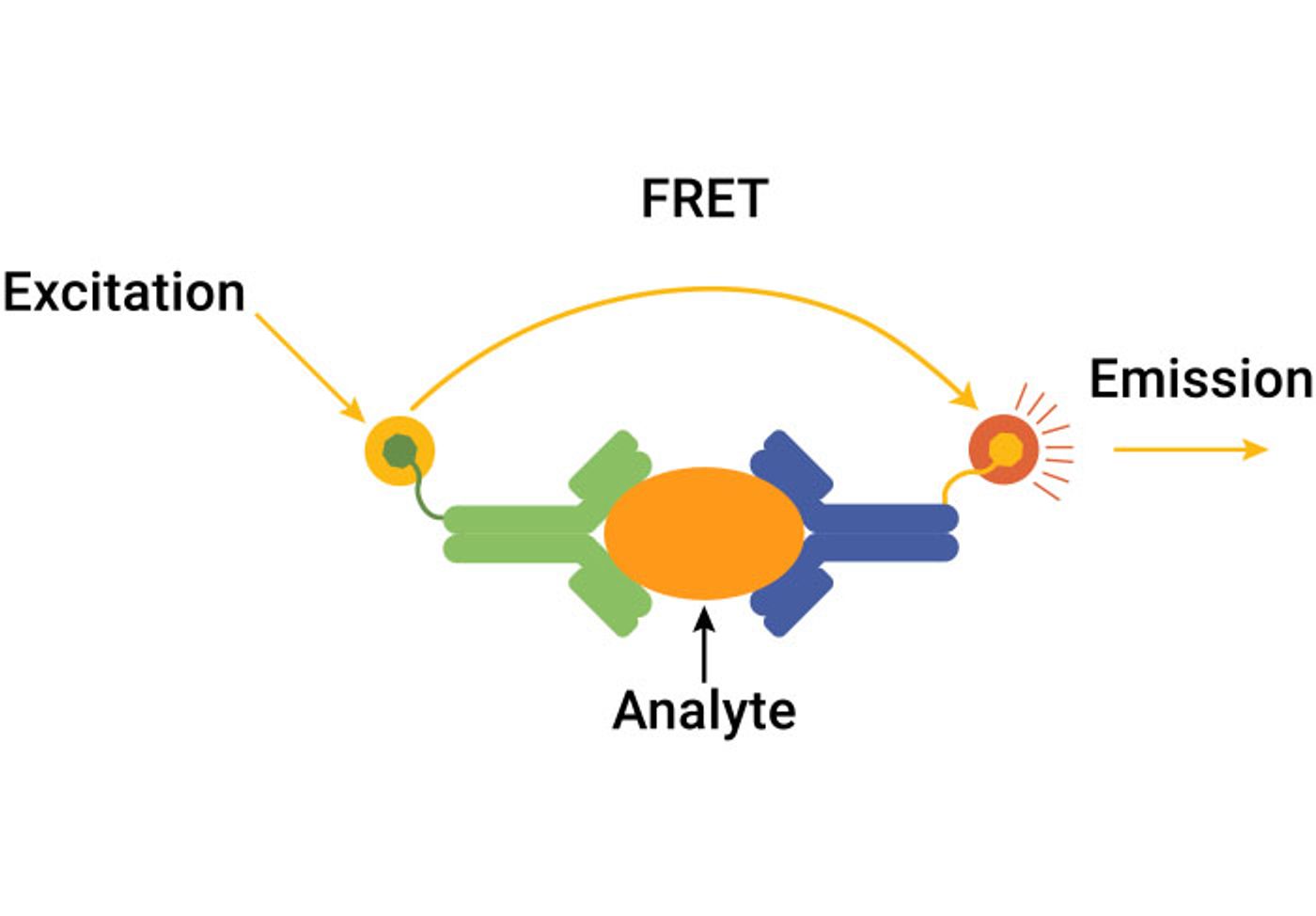
FRET-Based Immunoassays
Fluorescence resonance energy transfer (FRET) immunoassays are based on energy transfer between a donor fluorophore and an acceptor fluorophore. The fluorophores are conjugated to antibodies that are brought into proximity when bound to target analyte. The donor fluorophore must be excited by an energy source such as a lamp or laser, which requires specialized instrumentation. Both the donor and acceptor fluorescence are measured, and results are generated as a ratio of the two signals. Variations of FRET-based immunoassays include time-resolved fluorescence (TR-FRET) and HTRF® (homogenous time resolved fluorescence) that uses an alternate donor molecule to reduce background signal and increase signal stability.
- Benefits: Homogenous assay format, scalable to 384-well format
- Limitations: Requires specialized instruments; must be performed in low-light environment
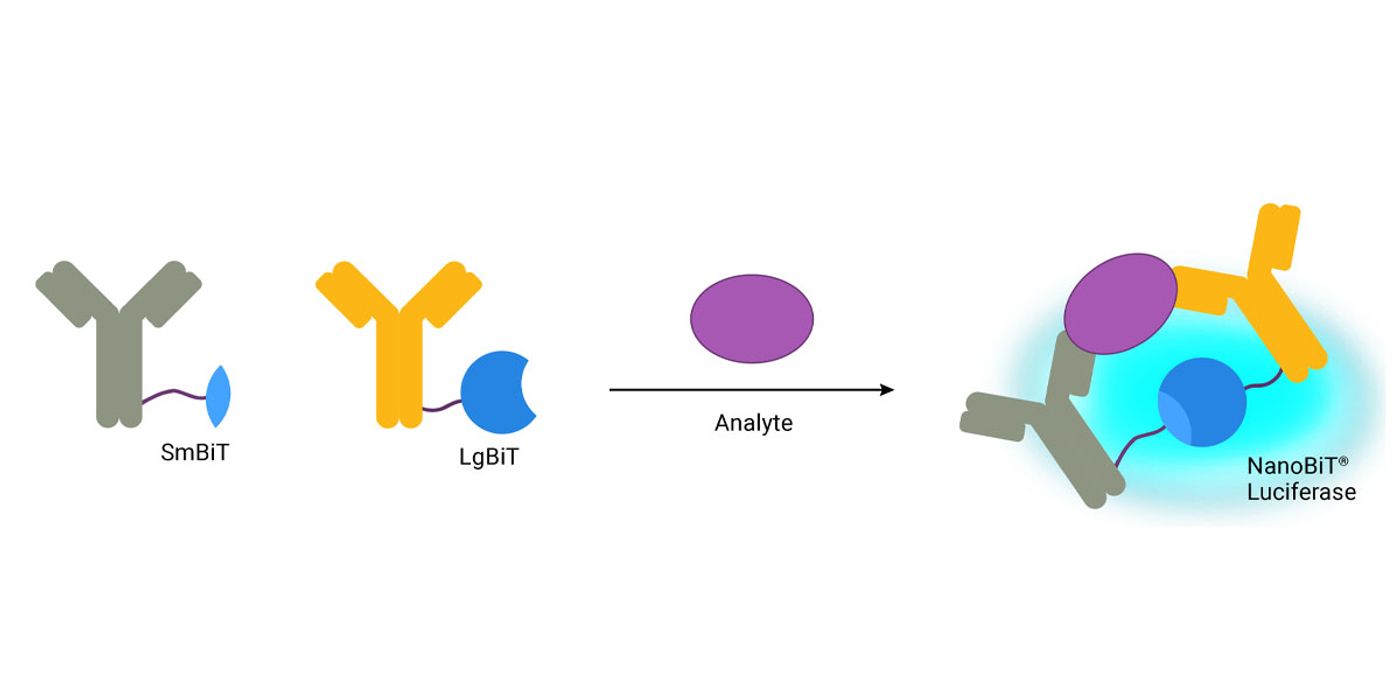
Lumit™ Immunoassays
Lumit™ Immunoassays are based on NanoBiT® technology—a protein complementation system that uses two subunits of an engineered luciferase enzyme: small bit (SmBiT) and large bit (LgBiT). These two subunits have low affinity for each other and can only form an active NanoLuc® luciferase when in close proximity. To detect a specific antigen, antibodies are labeled with LgBiT and SmBiT and mixed with sample. As the antibodies bind to the antigen, LgBiT and SmBiT are brought close together to form functional NanoLuc® luciferase. A detection reagent containing substrate is added to generate a luminescent signal that is proportional to target antigen levels. The simple add-mix-read protocol requires no wash steps and can be completed in as little as 30–90 minutes. Everything is performed in-well, enabling direct analyte measurement within the cell culture plate or on medium removed from the cells, making them ideal for high-throughput applications.
- Benefits: Fast; no wash steps; high sensitivity; wide dynamic range; does not require precoated plates or specialized instruments
- Limitations: Not all assays are compatible with serum samples
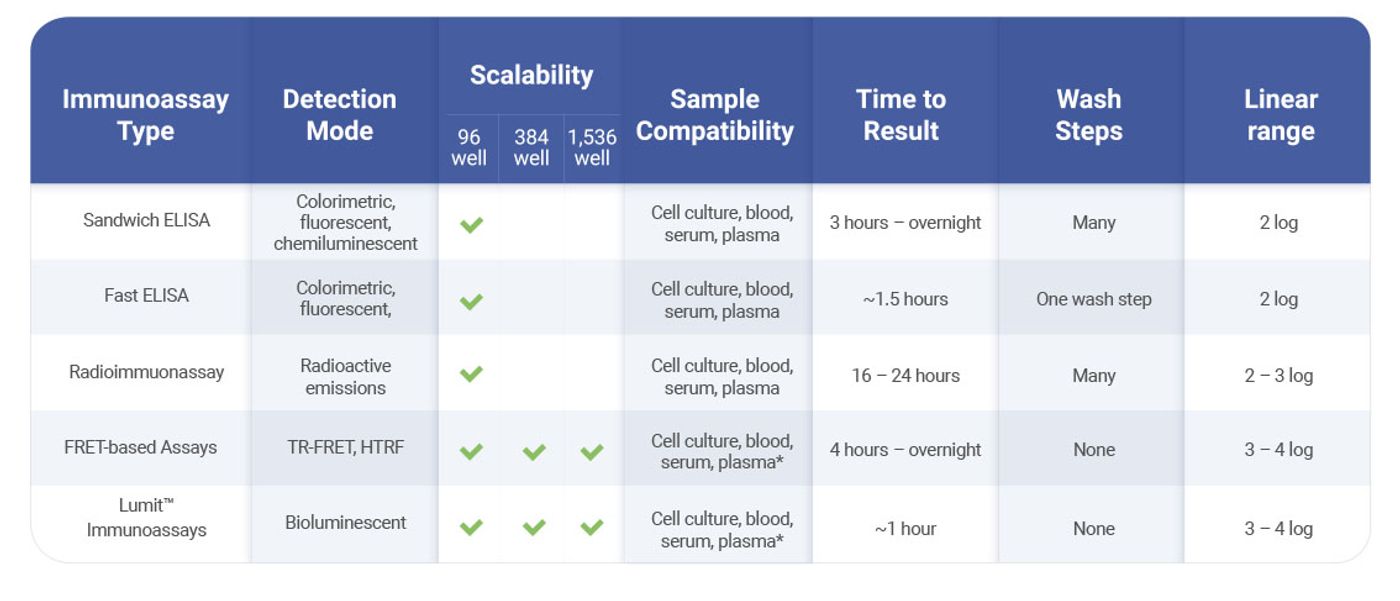
Considerations for Selecting an Immunoassay
With all the available options, selecting the right immunoassay for your purpose can be a daunting task. Below are some questions to consider when choosing an immunoassay:
-
What Instruments Do You Have Access To?
Many commercial immunoassay kits require specialized instruments for detection and plate-washing, which can be expensive. If your lab is on a tight budget and does not have access to these instruments, these kits may not be the best choice. In this case, you may want to choose immunoassays that can use fluorescence or luminescence detection, as they only need standard plate readers for detection.
-
How Many Samples Do You Have?
If you are routinely processing hundreds or thousands of samples, it is important to find an immunoassay that can scale up to fit your needs. Some assays are limited to 96-well formats, while others are compatible with 1,536-well plates.
-
What is Your Sample Type?
Most immunoassays can be used with cell culture samples. If you are testing other samples, such as blood or serum, it is important to choose an immunoassay that is compatible with your specific sample type.
-
How Fast Do You Want Results?
The time it takes to complete an immunoassay can vary drastically—some require overnight incubations, while some assays can be completed in just 1 hour. In addition, assays that require no wash steps will allow more time for you to focus on other research activities. Be sure to evaluate the cost-benefit ratio; sometimes it is worth the extra cost to save hours or days of work in the long run.
-
What Sensitivity is Required?
The sensitivity of an immunoassay is important if you want to ensure detection of antigens at low concentrations. Assays with a wider dynamic range are ideal to get accurate measurements of antigen from very low to very high concentrations and avoid the inconvenience of diluting samples before measuring.
*Sample compatibility is dependent on individual assays.