Strand of RNA Shuts Down Deadly Pediatric Cancer
A new pediatric brain cancer treatment uses a short RNA strand to shut down a deadly mutation. Introducing into a cell an exact match for a mutant protein’s mRNA, called an antisense oligonucleotide (ASO), stops cancerous protein production before it starts.
Image Credit Tdvorak, Creative Commons license CC BY 3.0
Scientists have developed ASOs pharmacological projects since the early 1990s. One of the amazing benefits of these RNA treatments is their specificity for their RNA complement. This helps decrease the chance of unintended interactions with other cellular parts.
Zhang et al. from Cold Spring Harbor Laboratories wielded ASO therapy against a type of cancer custom-made for this approach, a tumorous cancer called diffuse midline gliomas (DMG).
In 2012, a different group of researchers discovered that up to 70% of all DMGs stemmed from a tumor-sparking mutation called an oncogenic event. Switching a single amino acid for a histone protein from a lysine to a methionine lets slip the translation of cancer-related genes. This mutation is called H3.3 K27M.
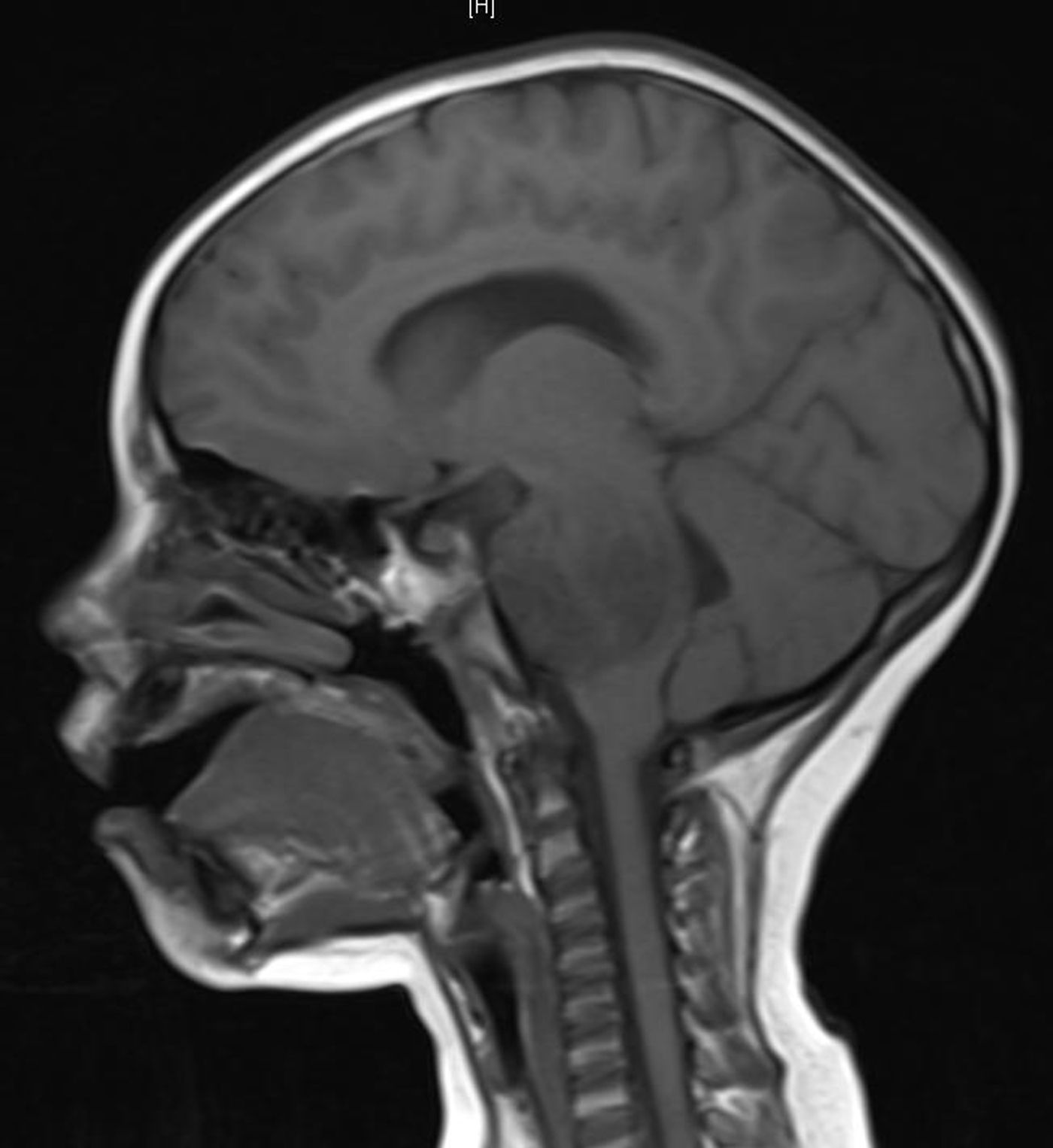
Around 20% of all pediatric central nervous system (CNS) tumors are DMGs. Grown rapidly deep within the brain, the two-year survival rate is less than 10% with most patients succumbing within 9 to 12 months after diagnosis.
The researchers tested their ASO treatment using DMG mouse models and tissue collected from deceased human patients. One model tested, called xenograft, involved taking samples from lethal tumors, processing them down to individual cells and injecting them into the fluid-filled spaces in the experimental mice brains.
Treatment with ASOs restored the growth of neural stem cells and crucial glial cells. The animals survived longer with slower tumor growth.
Sadly, the truth is until a delivery method is developed, the best treatments won’t get where they’re needed. The brain is so important that it’s encased in a skull of solid bone. Cancer-fighting drugs, including ASOs, cannot pass from the blood to the brain across the blood-brain barrier (BBB) without invasive delivery methods. This greatly contributes to failed DMG drug clinical trials.
One trial is currently underway to test the ability of a drug to cross the BBB to provide treatment for DMG (NCT03696355). Unfortunately, verification still requires a biopsy sample of the tumor. Another clinical trial attempts to focus low-frequency ultrasound waves on the brain to disrupt the BBB enough to allow a drug treatment to slip into the brain (NCT05123534).
Unfortunately, this new ASO treatment will be left on the shelf until delivery methods are discovered.
Sources: EurekAlert!, Science Translational Medicine, Nature reviews Neurology, NIH, Diagnostics, National Cancer Institute, Nature