Could Rotifers be the Next Great Laboratory Model?
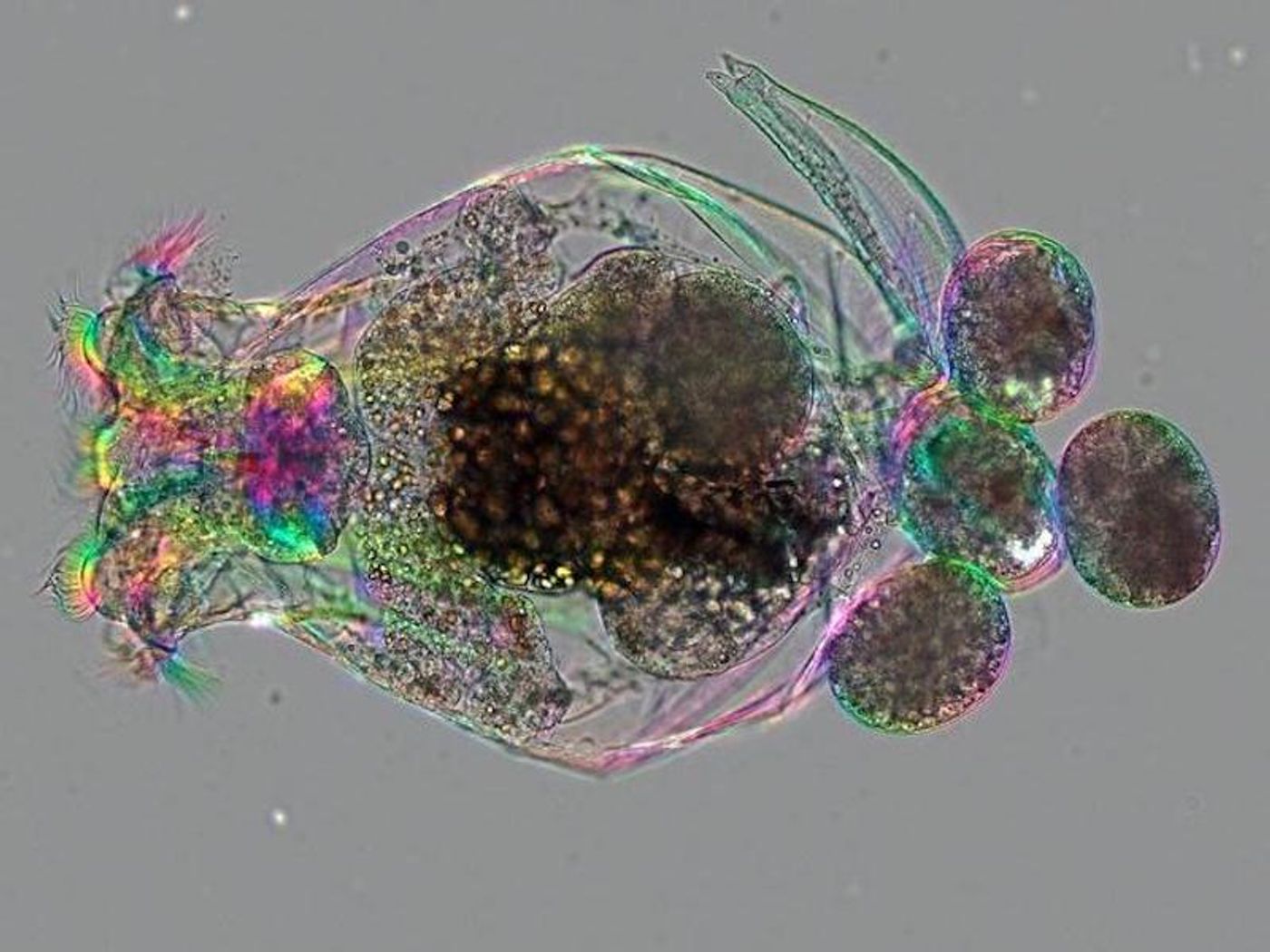
Rotifers are multicellular, microscopic marine animals that live in soils and freshwater environments. They are transparent and can be easily grown in large numbers. As such, they have been used in some laboratories as research subjects for many years. Now scientists have found a way to manipulate the rotifer genome, which can make them far more useful for many different research applications.
In new work reported in PLOS Biology, scientists used the CRISPR-Cas9 gene editing tool to alter two rotifer genes. These edits were then passed down to future generations of rotifers. This effort can now help others use these organisms in their laboratories.
"Our method turns out to be a very practical way to generate a large number of genetically altered rotifers fairly quickly," said co-corresponding study author David Mark Welch, a senior scientist at the Marine Biological Laboratory (MBL), among other appointments.
There are a variety of model organisms routinely used in laboratories, such as E. coli, yeast, fruit flies, zebrafish, mice, and rats. But these organisms cannot represent all the diversity found in biology, and do not always faithfully replicate human biology. Additional model organisms can provide fresh insights. Rotifers provide other benefits as well; since they are transparent, their development can easily be visualized; they are inexpensive to raise and easy to care for. They also have close connections to the ancestors of modern animals, so they are relevant. But ideally, it should also be relatively straightforward to manipulate the genomes of model organisms.
Since rotifers are very small - the largest of them can only just be seen with the naked eye - it's a challenge to deliver CRISPR gene editing reagents into the cells of these animals. They move around a lot, so researchers had to first find a way to keep them immobilized. They did so by immersing the rotifers in a thick solution, and exposing them to low levels of anesthesia. The investigators were then able to grasp the animals individually with a hollow needle, while light suction was applied.
After the animals are in place, the gene editing reagents can be delivered to the portion of the rotifer where nutrients are moved to eggs. Offspring that are hatched later from these eggs then carry the intended gene edits, and the researchers determined that these were passed onto the next generation.
In this study, the researchers deactivated a gene involved in development, called vasa. These animals stopped reproducing within a few generations. Another gene called mlh3 was also turned off or edited in a specific way, and either approach halted the production of male offspring. Now, the study authors are moving on to using rotifers in research that is more specific to their interests.
"This new tool, along with the ease of raising rotifers in the lab, will make it possible to use them to answer lots of questions we aren't even thinking about yet," said co-corresponding study author Kristin Gribble.
Sources: Marine Biological Laboratory, PLOS Biology