Gut Microbes Swap Resistance Genes Even More Easily Than We Knew
Antibiotics are often used to eliminate tough infections. Bacteria can easily exchange genetic information in a process called horizontal gene transfer. This enables microbes to share genes that encode for antibiotic resistance, which creates more pathogenic microbes that are difficult to get rid of when they cause illness. But researchers have now discovered that bacteria can swap genetic information even more readily than we thought. They use super polymer structures called F-pili to do so, and while this is very helpful to bacteria, researchers may be able to take advantage of the process to develop methods for drug delivery. The findings have been reported in Nature Communications.
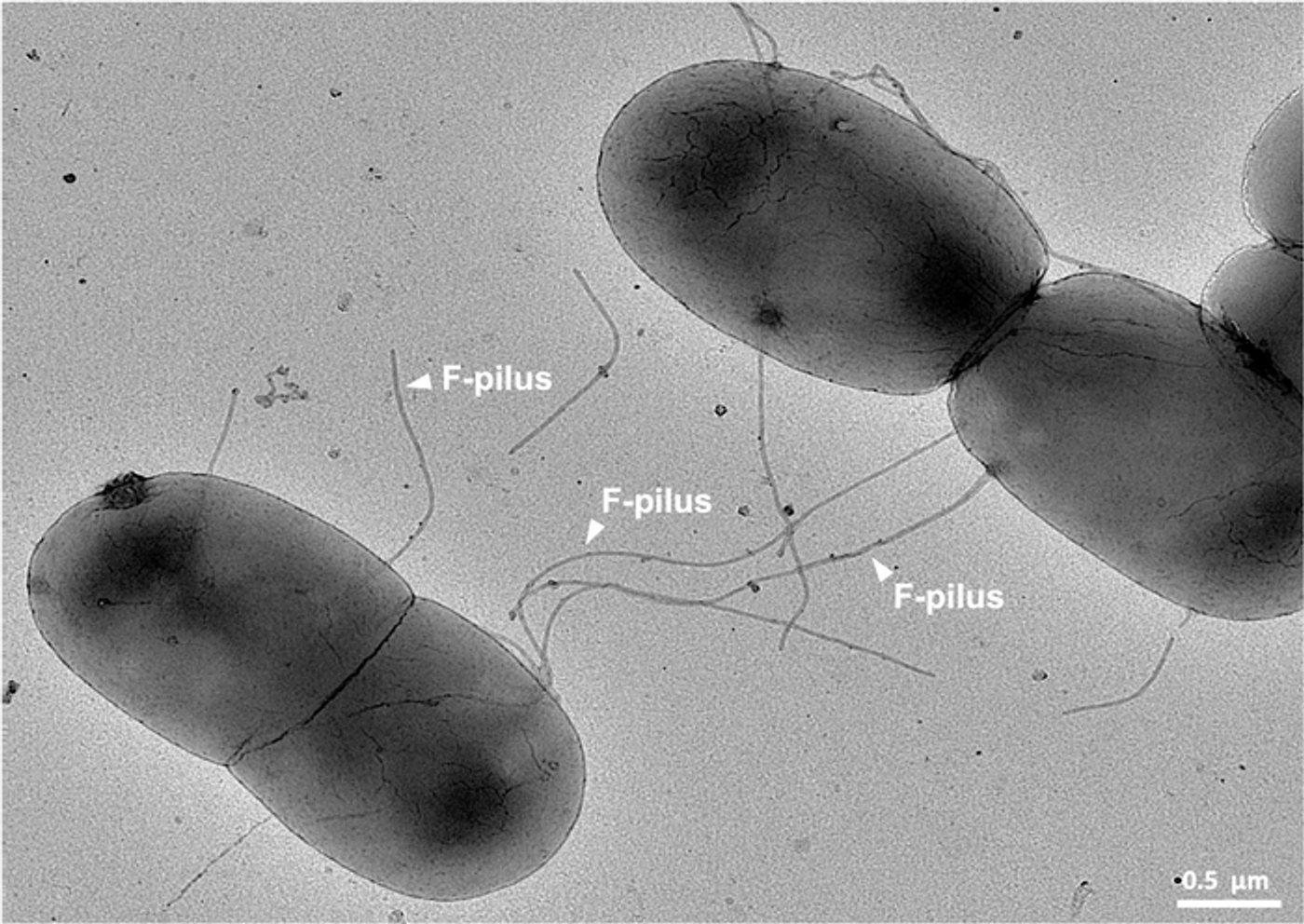
Gut microbes can form appendages known as F-pili that link the cells to one another and move antibiotic resistance genes. It was once thought that the gastrointestinal tract was a harsh environment that would disrupt or destroy F-pili. This work has shown otherwise, however. Not only are they not harmed, the F-pili actually seem to get stronger in these conditions, promoting the efficient movement of resistance genes, and gathering the microbes into biofilms.
Microbial biofilms can take on many powerful characteristics that individual microbial cells don't have; they are more difficult to get rid of and can get a tenacious hold on various surfaces.
Antibiotic-resistant infections are projected to cause as many deaths as cancer by the year 2050, noted first study author Jonasz Patkowski from the Department of Life Sciences at Imperial College London. Studies like this may help us reduce the number of resistant pathogens or find ways to stop them.
The researchers wanted to challenge the assumption that the gut is too harsh for F-pili, so they grew E. coli that used F-pili to link together, or conjugate. When the microbes were agitated by shaking, there was an increase in gene transfer among the microbes. These shaken bacteria were also able to form biofilms more easily, shielding them from antibiotics.
Additional work showed that the F-pili were highly elastic, and could spring back when stretched instead of breaking. The appendages were also tested under other conditions like those in the gut, including exposure to high temperatures and chemicals like urea or sodium hydroxide. The F-pili survived these conditions, and were still functional.
The investigators determined that the F-pili are composed of subunits made of interlinked phospholipids. These phospholipids are essential to F-pili. When F-pili lacking phospholipids were stretched, they lost their springiness and broke apart.
"Making F-pili is very costly to the bacteria in terms of resources and energy, so it's no surprise they are worth the effort," said senior study author Dr. Tiago Costa of the Department of Life Sciences at Imperial. "We have shown how F-pili accelerate the spread of antibiotic resistance and biofilm formation in turbulent environments, but the challenge now is to find ways to combat this very efficient process."
Sources: Imperial College London, Nature Communications