A Bacterial small RNA That Regulates Infection
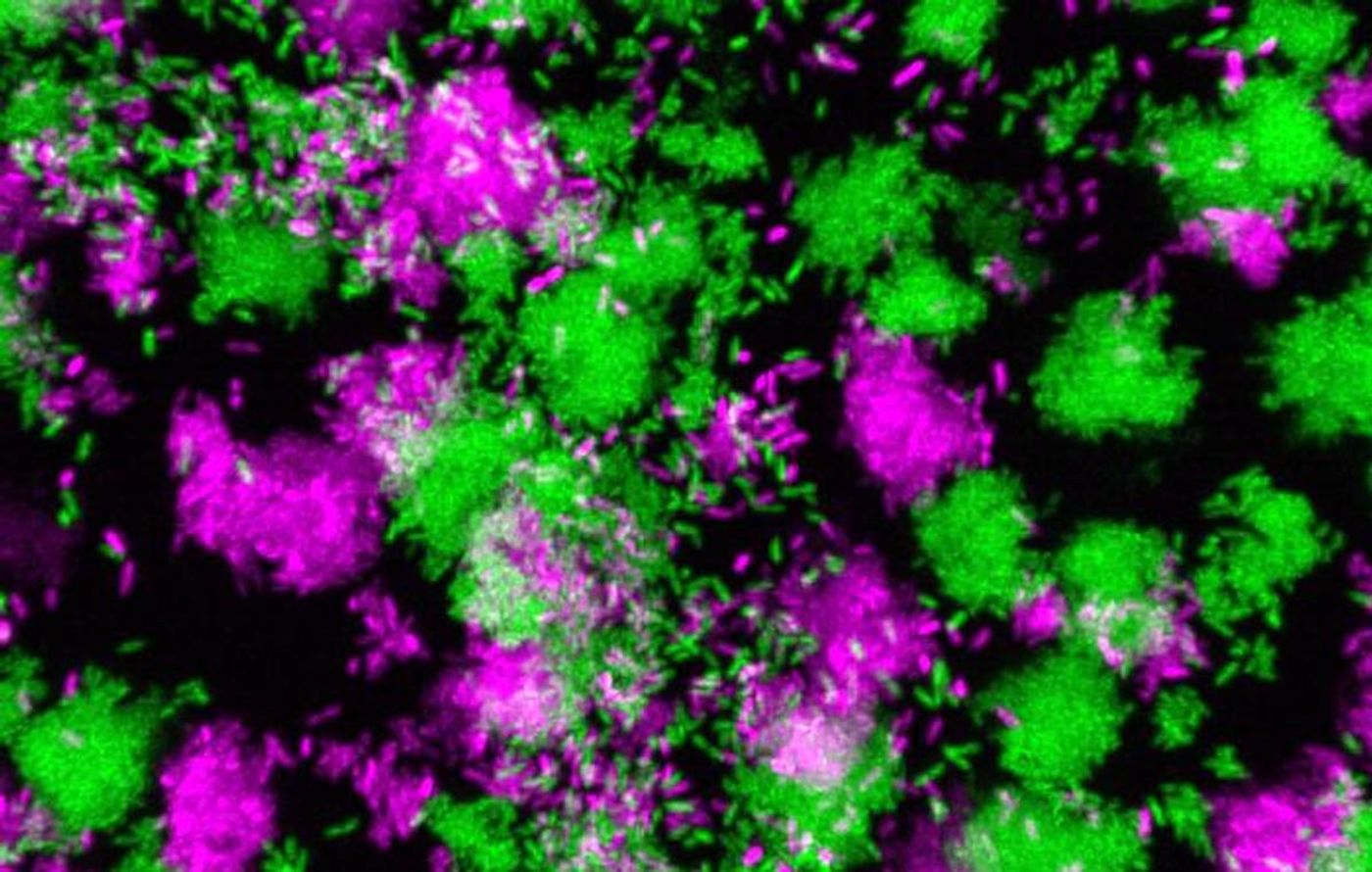
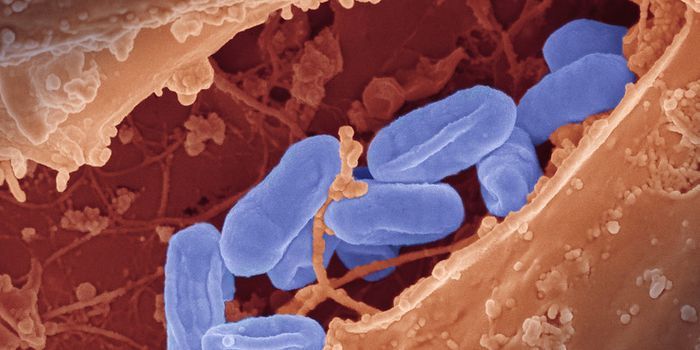
Infection is a constant risk. Any germs that make it past the body's barriers, like the skin, can cause illness if the immune system does not respond properly and efficiently. Most people can easily fight off common bacteria that could be pathogenic, but people with weaker or compromised immune systems have a greater risk of infection. For the immunocompromised, bacteria like Pseudomonas aeruginosa that are everywhere in the environment can start to grow in different parts of the body, including the lungs; if this occurs, a chronic and even lifelong infection can arise. If bacteria that cause these chronic infections move to the bloodstream, they can become far more serious and even deadly. Scientists are still learning about how bacteria are able to make the transition.
New work has provided insight into how P. aeruginosa can change from causing one type of infection to another. This study used human samples to reveal a biomarker that indicates when this transition has taken place, in the form of a small RNA generated by the bacterium. The findings have been reported in Nature.
Instead of growing bacteria in the lab, the researchers assessed the bacteria within a human host. They identified all of the genes that the infectious microbes were expressing by sequencing all of the bacterial mRNA.
There are about 6,000 genes in the P. aeruginosa genome, and one in particular called PA1414 was expressed in human tissues at higher levels than every other bacterial gene combined. This excessive level of expression initially led the investigators to believe there was a problem with their methods.
"This particular gene is not expressed in the standard lab environment very much, so it was striking to see these levels. And at this point, the function of the gene was unknown," said first study author Pengbo Cao, a postdoctoral researcher in the lab of Professor Marvin Whiteley at the Georgia Institute of Technology.
This gene, small RNA SicX (sRNA inducer of chronic infection X), does not encode for any proteins, either. Instead, it appears to be involved in bacterial respiration. Low oxygen levels were found to promote expression of this gene, which is a common part of bacterial infections. Bacteria are usually deprived of oxygen when a chronic infection is underway.
When SicX was eliminated in an animal model of infection, the bacteria quickly moved from chronic infections to cause systemic illnesses. Thus, the gene seems to promote chronic, local infections. When bacterial infections changed from chronic to acute, SicX expression dropped, suggesting that the gene could be a biomarker for this transition.
"In other words, without the small RNA, the bacteria become restless and go looking for oxygen, because they need to breathe like we need to breathe," Whiteley said. "That need causes the bacteria to enter the bloodstream. Now, we know that oxygen levels are regulating this transition."
If clinicians can tell when an infection will change, treatments may improve, along with patient outcomes. A diagnostic test could tell people when treatment is necessary and before their life is threatened, added Whitely, who is corresponding study author.
"The chronic Pseudomonas infection is usually highly resistant to first-line antibiotics," Cao added. "By targeting this small RNA, we could potentially change the lifestyle of the bacteria to make it more susceptible to antibiotic treatments and achieve greater clearance of these dangerous infections."
Sources: Georgia Institute of Technology, Nature