New Hope for Epilepsy Patients: Experimental Regenerative Brain Cell Therapy
Doctors at UC San Diego Health recently performed a clinical trial surgery involving the injection of regenerative cells into a patient’s brain that could be used to treat epileptic seizures. This surgery marks just the third time such a procedure was performed and is a part of a national clinical trial designed to use human stem cells to provide epilepsy patients with a non-destructive option for those whose temporal lobes exhibit drug-resistant characteristics.
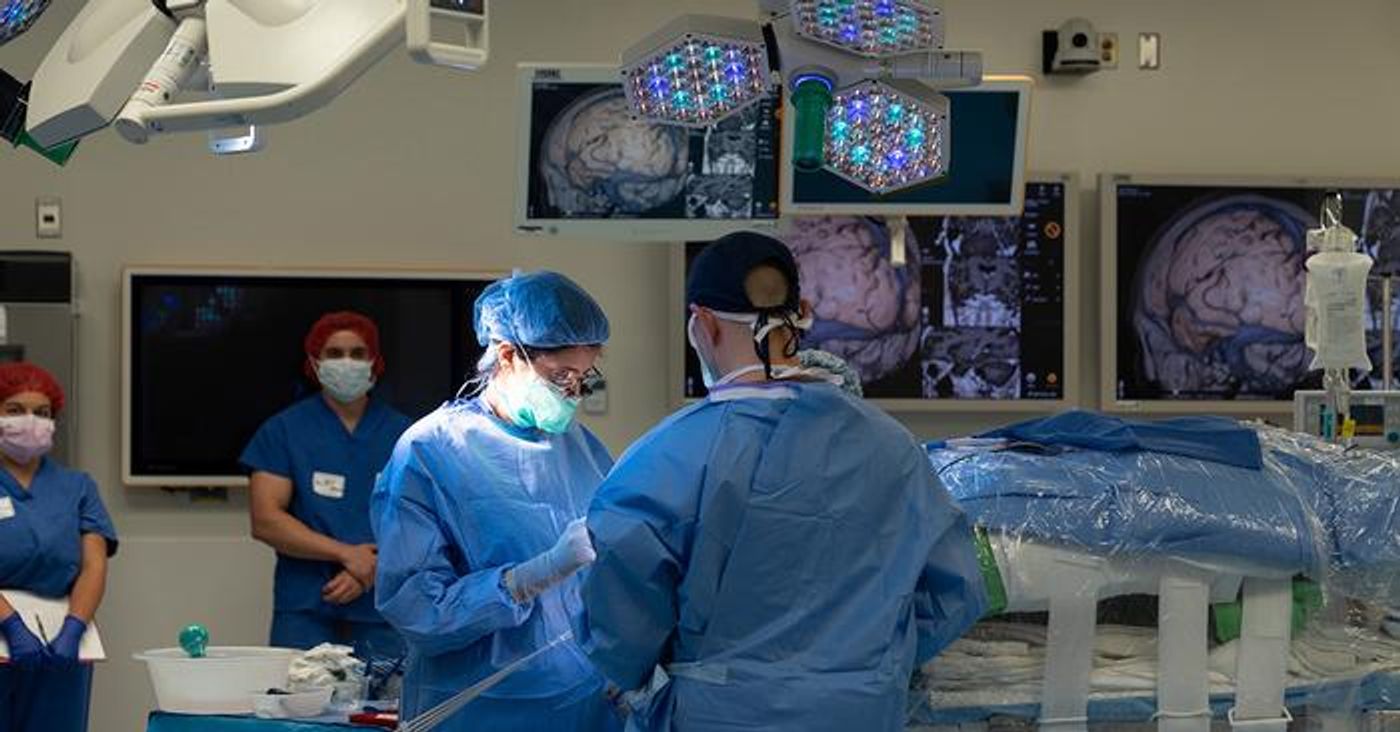
Dr. Sharona Ben-Haim, MD, seen performing only the third ever experimental regenerative brain cell therapy for epilepsy in July 2023, which is part of a national clinical trial designed to eliminate seizures, thus revolutionizing the treatment of temporal lobe epilepsy. (Credit: UC San Diego Health)
“This experimental therapy offers us the potential to essentially restore the balance in the brain to be able to calm and ideally stop the seizures, while retaining the normal function of that part of the brain,” said Dr. Sharona Ben-Haim, MD, who is an associate professor of neurological surgery at UC San Diego School of Medicine, and who performed the experimental procedure. “Currently, we do not have a therapy that allows us to do that, so this is really exciting.”
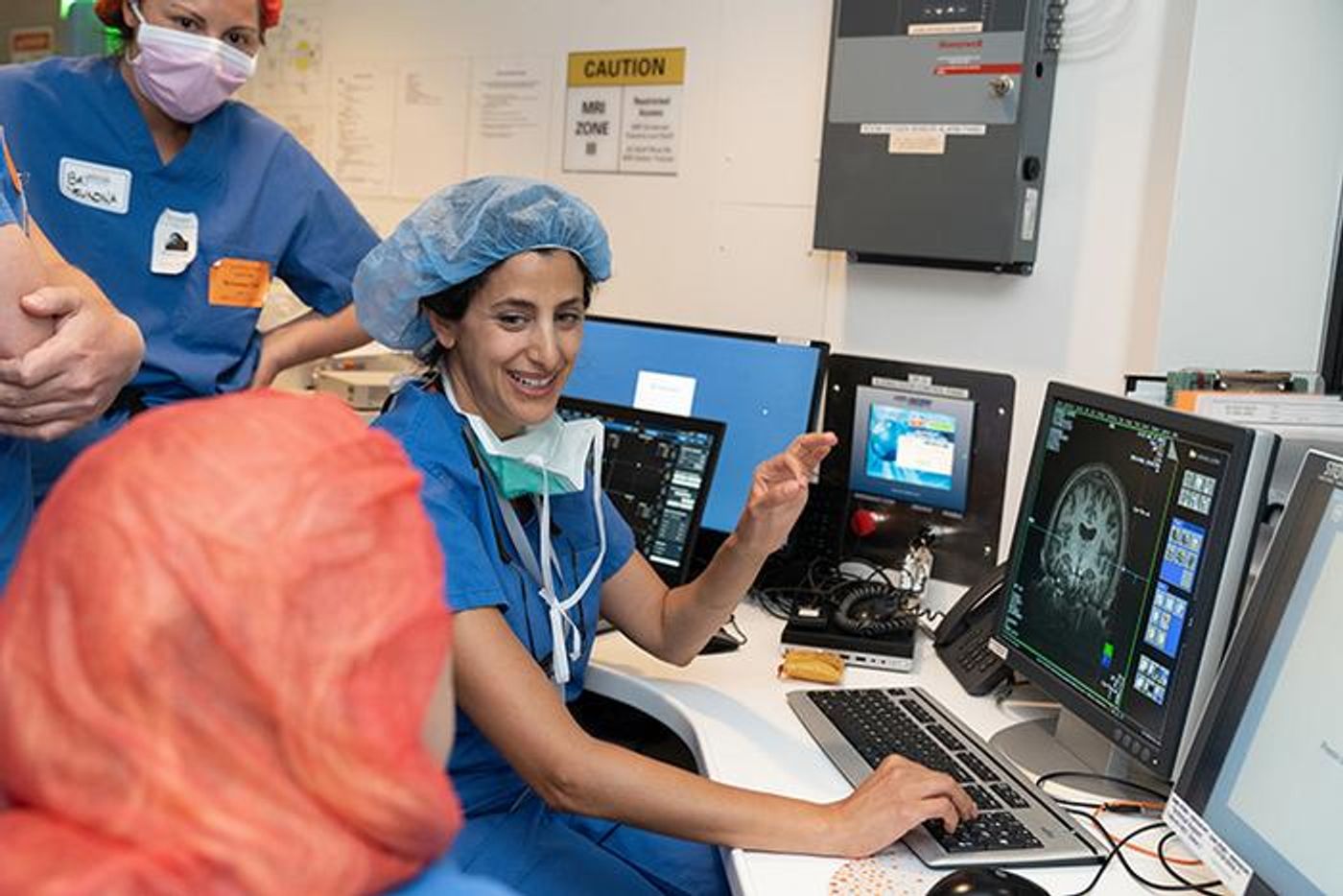
During the procedure, Dr. Ben-Haim carefully examined the magnetic resonance imaging (MRI) scans of the patient’s brain to navigate where to inject the human stem cell-derived inhibitory brain cells, also called interneurons. The stem cells are designed to create what’s known as gamma-aminobutyric acid (GABA) which is a commonly known neurotransmitter that inhibits hyperactive impulses that might occur between brain cell nerves and is known for reducing both stress and anxiety while improving sleep. Sponsored by Neurona Therapeutics, the goal of the clinical trial to sign up 40 volunteers across the United States to take part in the study. After the procedure, Dr. Ben-Haim examined her work with the aid of a 3D digital image.
Dr. Sharona Ben-Haim, MD, with a 3D digital image of the procedure. (Credit: UC San Diego Health)
The Centers for Disease Control and Prevention (CDC) reports that approximately 3.4 million Americans—3 million adults and 470,000 children—suffer from epilepsy with about one-third being resistant to anti-seizure medications. Additionally, adults suffering from the disorder describe worse physical and mental health, along with financial difficulties from being unable to afford various forms of health care in contrast to adults without the disorder.
The World Health Organization reports that approximately 50 million people suffer from epilepsy around the world, with an estimated 75 percent of those individuals in low-income countries not receiving proper treatment. What makes this new procedure groundbreaking is its non-invasive method to treat epileptic seizures, since conventional surgical therapies involve laser-burning or removing the epileptic parts of the brain.
“This first-in-human clinical trial represents a paradigm shift in the way we treat this disease process, shifting from procedures that destroy bad tissue to procedures that repair the bad tissue,” said Dr. Jerry Shih, MD, who is a professor of neurosciences at UC San Diego School of Medicine, along with being a neurologist and director of the Epilepsy Center at UC San Diego Health. “Our hope is that this procedure has such a high success rate and good tolerability that it becomes the standard of care for all drug-resistant focal epilepsies.”
While this was only the third ever procedure of this type, initial results released in June 2023 indicate a 90 percent drop in seizure frequency for the first patient one year after surgery and seven months after surgery for the second patient. The goal is to monitor each patient for up to two years after the procedure.
What new discoveries will surgeons make about controlling epileptic seizures with this new procedure in the coming years and decades? Only time will tell, and this is why we science!
As always, keep doing science & keep looking up!
Sources: EurekAlert!, U.S. National Library of Medicine, UC San Diego Health, Cleveland Clinic, Neurona Therapeutics, Centers for Disease Control and Prevention, World Health Organization