Neurons "Borrow" Serotonin From Each Other to Drive Different Behaviors Across Time
Understanding the brain's intricate machinery and how it orchestrates behaviors is a long-standing challenge in neuroscience. Yet, how can we ever hope to understand complex behaviors such as depression when the human nervous system can support around 600 trillion synaptic connections, each neuron averaging around 7,000?
One way to take on this challenge is to research neurotransmission within a system with fewer neurons and restricted behavior, such as the roundworm C. elegans'. From a small number of 302 neurons, these worms are capable of a moderate range of categorized wiggly behaviors.
In a recent study conducted at MIT and published in the journal Current Biology, Yung-Chi Huang and fellow researchers show that one type of neuron can produce multiple behaviors through a surprising mechanism.
The team focused on a specific neuron type known as the Hermaphrodite-Specific Neurons (HSN). Linked to motor control, HSN is thought to initiate the egg-laying behavior of female C. elegans. Previous research hinted that serotonin and other neuropeptides were involved in HSN-induced egg-laying.
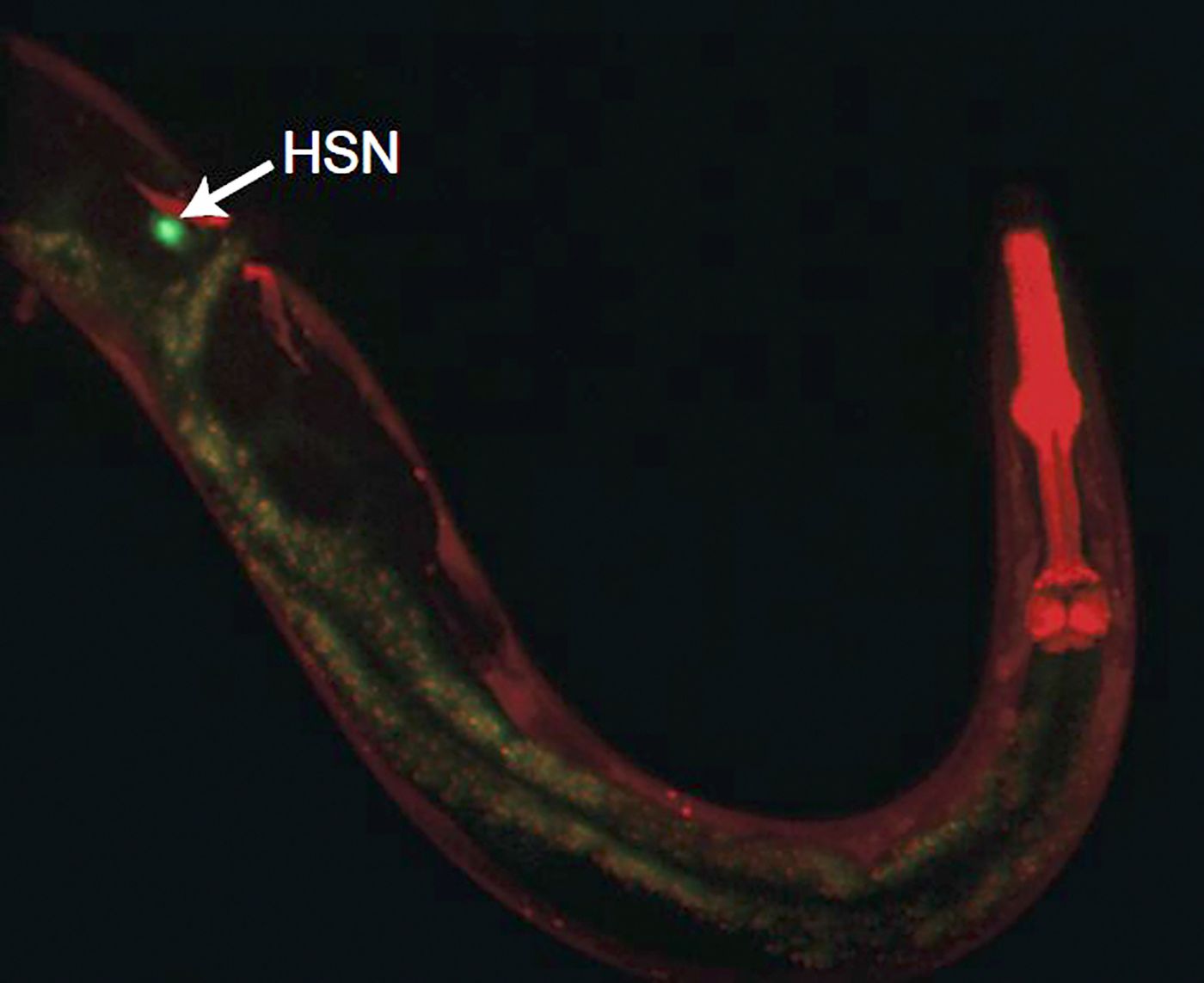
Flavell Lab/MIT Picower Institute
In a C. elegans worm the neuron HSN has a critical role in directing several coordinated behaviors. This image labels HSN in green, showing its extension all the way to the worm's head (labeled in red).
Huang et al. discovered that HSN exhibits remarkable flexibility, promoting egg-laying and the burst in speed that occurs concurrently and serves to disperse the eggs across nutritionally dense areas. After strafing the landscape, HSN initiates slowing down and dwelling behaviors, presumably so the worm could also enjoy the resource-rich real estate. The same cells mediate diverse behaviors by utilizing multiple transmitters in distinct cellular regions.
This revelation challenges the traditional view of neurons having fixed roles and emphasizes their versatility in driving various behaviors over different timescales. The study also shed light on interactions between neurons, uncovering a fascinating process where serotonin released by HSN is "borrowed" between neurons. A separate cell type, NSM neurons, were recorded taking up the serotonin released by HSN and re-releasing it to drive dwelling behavior directly.
These findings open exciting avenues for further exploration into how neurons use neurotransmitters to shape behaviors. Many researchers studying C. elegans' serotonin signaling hope that learning more about the fundamentals of serotonin signaling will aid in understanding and treating mood disorders such as depression, which physicians commonly manage with drugs that adjust neurotransmitter activity.
The findings unveiled the diverse array of functions that a solitary neuron type can fulfill. We now have valuable insights into the dynamic ways neuronal signals translate into behavior.
Sources: Current Biology, The Jackson Laboratory, PLoS Genetics, EurekAlert!