Accelerating Diagnostics Redirect
- Lateral flow design considerations
High performance, diagnostic rapid point-of-care (POC) flow-based tests can quickly deliver reliable results in challenging settings. When designing a new test and designing out a component in an existing test-what are the key considerations and what will be needed in the future?
- Accelerating diagnostic development with custom and design services
Commercializing a test or assay as quickly as possible is critical to success. Find out how diagnostic design services, lyophilization and custom capabilities can provide capacity, resources and expertise to improve speed to market now and in the future.
- Collaborating for effective diagnostic development
Strategic collaboration enables the development of diagnostic tests, assays and services for a wide range of applications. Hear about recent collaborations that have resulted in successful test development and set a path for future development.
- Simplifying your next-generation sequencing (NGS) workflow
High throughput NGS techniques are revolutionizing genomics from massively parallel whole genome sequencing to targeted sequencing approaches. Find out how to simplify your sequencing approaches for more detailed data, better diagnosis, and more targeted treatments to address current and future demands.
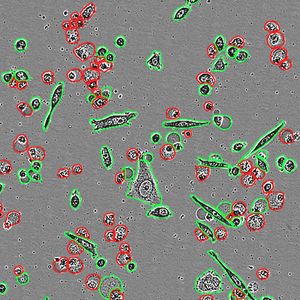
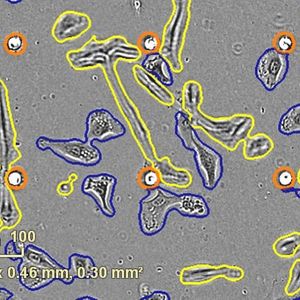
- Exploring single-cell omics opportunities
Recent technological advancements have made single-cell omics an increasingly powerful tool for diagnosis, treatment response prediction, and treatment selection, particularly for cancer. Learn about the opportunities for improving results in this growing field.
- Optimizing nucleic acid isolation
Recent years have seen the development and adaptation of several approaches for nucleic acid isolation, aiming to improve speed, throughput, and reproducibility. Find out how to optimize results, particularly for liquid biopsy and viral samples now and in the future.
Sessions will be available for on-demand viewing immediately following the live broadcast.
Advancing the development and manufacture of therapeutics
Cytiva is a global life sciences leader dedicated to advancing and accelerating therapeutics. Cytiva is a trusted partner to customers that undertake life-saving activities ranging from biological research to developing innovative vaccines, biologic drugs, and novel cell and gene therapies. Cytiva brings speed, efficiency and capacity to research and manufacturing workflows, enabling the development, manufacture and delivery of transformative medicines to patients. Visit cytiva.com for more.
Improving access to life-changing therapies that transform human health
Our customers undertake life-saving activities ranging from fundamental biological research to developing innovative vaccines, biologic drugs, and novel cell and gene therapies. Our job is to supply the tools and services they need to work better, faster and safer, leading to better patient outcomes.
Cytiva is a global life sciences leader with over 7000 associates across 40 sites who are dedicated to our vision to improve access to life-changing therapies that transform human health. As a trusted partner to customers that range in scale and scope, Cytiva brings efficiencies to research and manufacturing workflows, ensuring the development, manufacture and delivery of transformative medicines to patients.
- We operate in 40 countries and territories covering Europe, Asia, North and Latin America, and the Middle East.
- 7,000 associates are dedicated to our mission to advance and accelerate therapeutics.
- We host 11 customer-centered innovation facilities across Asia, Europe and the Americas to carry ideas through to commercial development
Find out more at Cytiva.com
We are delighted to be hosting a Panel Q&A session at the Diagnostics virtual open day on 17 June 2021.
If you have questions on diagnostic test development, we want to hear from you. Submit your questions to our team of experts on topics including lateral flow assay development, diagnostic design and custom services and optimizing sample preparation and purification with magnetic beads...
We will do our best to answer as many of the questions as possible during the open day.
Submit your questions through www.slido.com enter code #DXOPENDAY
We have also compiled the most popular questions that were submitted by attendees at the Diagnostics Virtual Summit in October 2020 which we will also answer during the event.
Speakers Share
-
Anja Drescher
Senior Application Specialist for Biacore systems, Cytiva
BIOGRAPHY
-
Gabriel Fernandez de Pierola
General Manager, Genomics & Diagnostic Solutions, Cytiva
BIOGRAPHY
-
Dr. Chris Fronczek
Application Scientist, BioDot, Inc
BIOGRAPHY
-
Priya Hari
Post Doctoral Genomics Research Fellow, Cytiva
BIOGRAPHY
-
Klaus Hochleitner, PhD
Global Technology Lead, Diagnostics, Cytiva
BIOGRAPHY
-
Lee Jenkins
Senior Product Leader, Cytiva
BIOGRAPHY
-
Marilena Loizidou
Deputy Director Division of Surgery and Interventional Science, University College London (UCL) ...
BIOGRAPHY
-
Tom Lyons
Custom Projects Leader, Genomics and Diagnostic Solutions, Cytiva
BIOGRAPHY
-
Sean Mackay
CEO & Co-Founder, Isoplexis
BIOGRAPHY
-
Pieter Mestdagh, PhD
Senior Scientist at Biogazelle and Associate professor, Faculty of Medicine and Health Sciences, Ghent University, Belgium
BIOGRAPHY
-
Dr. Zahidul Mondle
Lead Technical Product Specialist, Cytiva
BIOGRAPHY
-
Chris Norey
R&D Leader, Genomics and Cellular Research, Cytiva
BIOGRAPHY
-
Brendan O'Farrell, PhD
President and Co-Founder, DCN Diagnostics
BIOGRAPHY
-
David Raw
Supplier Risk Management Leader, Cytiva
BIOGRAPHY
-
Darren Rowles
President and CEO, Sona Nanotech
BIOGRAPHY
-
Monika Seidel
Senior Development Scientist - Genomics and Cellular Research, Cytiva
BIOGRAPHY
-
Stefanie Stepanow, Dr. rer nat
NGS Core Facility Manager, Private Molecular Diagnostic Lab, Cologne, Germany.
BIOGRAPHY
-
Jonathan Thomas
Head of Contract Sales, Abingdon Health
BIOGRAPHY
-
Amy Webster
Custom Projects Leader, Cytiva
BIOGRAPHY
-
David Wilson
Commercial Director, Diagnostics, Avacta Life Sciences
BIOGRAPHY
Abingdon Health is a technology-enabled lateral flow diagnostics company providing leading-edge rapid testing solutions to a multi-industry, global client base. Abingdon Health provides lateral flow assay development and smartphone reader solutions alongside Europe’s largest rapid test manufacturing capacity. A multi-disciplinary team forms the foundations of our services and a partnership-led approach alongside a focus on operational excellence produces reliable products irrespective of their complexity. Established supply chain and manufacturing processes are tailored to assay specifications and market demand for continuity of supply.
Avacta is developing novel cancer immunotherapies combining its two proprietary platforms – Affimer® biotherapeutics and pre|CISIONTM tumour targeted chemotherapy. With this approach, the Company aims to address the lack of a durable response to current immunotherapies experienced by most patients. The Company’s therapeutics development activities are based in Cambridge, UK. The Company benefits from near-term revenues generated from Affimer reagents for diagnostics, bioprocessing and research, through a separate business unit based in Wetherby, UK. The Affimer platform is an alternative to antibodies derived from a small human protein. Despite their shortcomings, antibodies currently dominate markets worth in excess of $100bn. Affimer technology has been designed to address many of these negative performance issues, principally: the time taken, and the reliance on an animal’s immune response, to generate new antibodies; poor specificity in many cases; large size and cost. Avacta’s proprietary pre|CISIONTM targeted chemotherapy platform, releases active drug only in the tumour, thereby limiting systemic exposure and improving the overall safety and therapeutic potential of these powerful anti-cancer treatments. Avacta expects to take its first pre|CISIONTM drug candidate, a targeted form of the standard-of-care Doxorubicin, into the clinic by the first half of 2021. By combining these two platforms the Company is building a pipeline of novel cancer therapies with the aim of creating effective treatments for all cancer patients including those who do not respond to existing immunotherapies. Avacta’s diagnostics business unit works with partners world-wide to develop Affimer proteins for evaluation by those third parties with the objective of establishing royalty bearing license deals with a particular focus on the diagnostics sector. The Company is also developing a small in-house pipeline of Affimer-based diagnostic assays for licensing.
BioDot is the leading provider of automated, ultralow-volume, non-contact fluid dispensing platforms for R&D applications and commercial-scale manufacturing across diagnostics and life sciences, medical products and applied industrial markets. Its technologies are integral to production of point-of-care diagnostics for infectious diseases, allergy tests and diabetes maintenance as well as veterinary medicine and environmental applications. BioDot, founded in 1994, is based in Irvine, California and has international offices in Europe and Asia.
Biogazelle is a Contract Research Organization (CRO) specializing in high-value applications to support pharmaceutical research, clinical trials and diagnostic test development. We hold a unique forefront position in the application of quantitative PCR, digital PCR and dedicated RNA sequencing workflows, optimized for quantification of coding and non-coding genes, on precious clinical samples such as liquid biopsies and formalin-fixed, paraffin-embedded (FFPE) tissues. Our aim is to accelerate the development of therapeutics, including small molecules, RNA targeted drugs, and adoptive cell therapies.
IsoPlexis (www.isoplexis.com) is dedicated to accelerating the fight against cancer and a range of the world's toughest diseases with its uniquely correlative, award-winning, single-cell proteomics systems. By revealing unique immune biomarkers in small subsets of cells, we are advancing immunotherapies and targeted therapies to a more highly precise and personalized stage. Our integrated systems, named #1 innovation by the Scientist Magazine and Fierce, are used globally to advance the field of immune biology and biomarkers as we generate solutions to overcome the challenges of complex diseases.